Report Author: Haidy Kamel
Report Date: March 21, 2025
Table of Contents
Introduction
The immune system continuously works to defend the body against harmful pathogens, relying on a consistent supply of essential nutrients from the diet to function optimally. In addition to combating infections, the immune system must maintain tolerance to avoid inappropriate responses against the body’s own healthy tissues or harmless foreign substances.
A crucial component of immune defense is the upper respiratory tract, which serves as the body’s first line of defense against airborne pathogens. This region includes the nose, mouth, nasal cavity, pharynx, and larynx, which work together to filter, warm, and humidify incoming air while preventing harmful particles from reaching the lungs. The trachea, though part of the lower respiratory tract, also contributes to immune defense through its mucociliary escalator—a mechanism that traps and removes pathogens and debris before they reach the lungs. The nasal cavity and trachea contain mucus-secreting cells and cilia (hairlike structures) that work to trap and expel harmful particles, while the pharynx and larynx aid in respiration and protection by directing air to the lower respiratory tract and preventing foreign material from entering the lungs (Figure 1). These anatomical structures, along with their immune-associated functions, are essential for maintaining respiratory health and preventing infections.
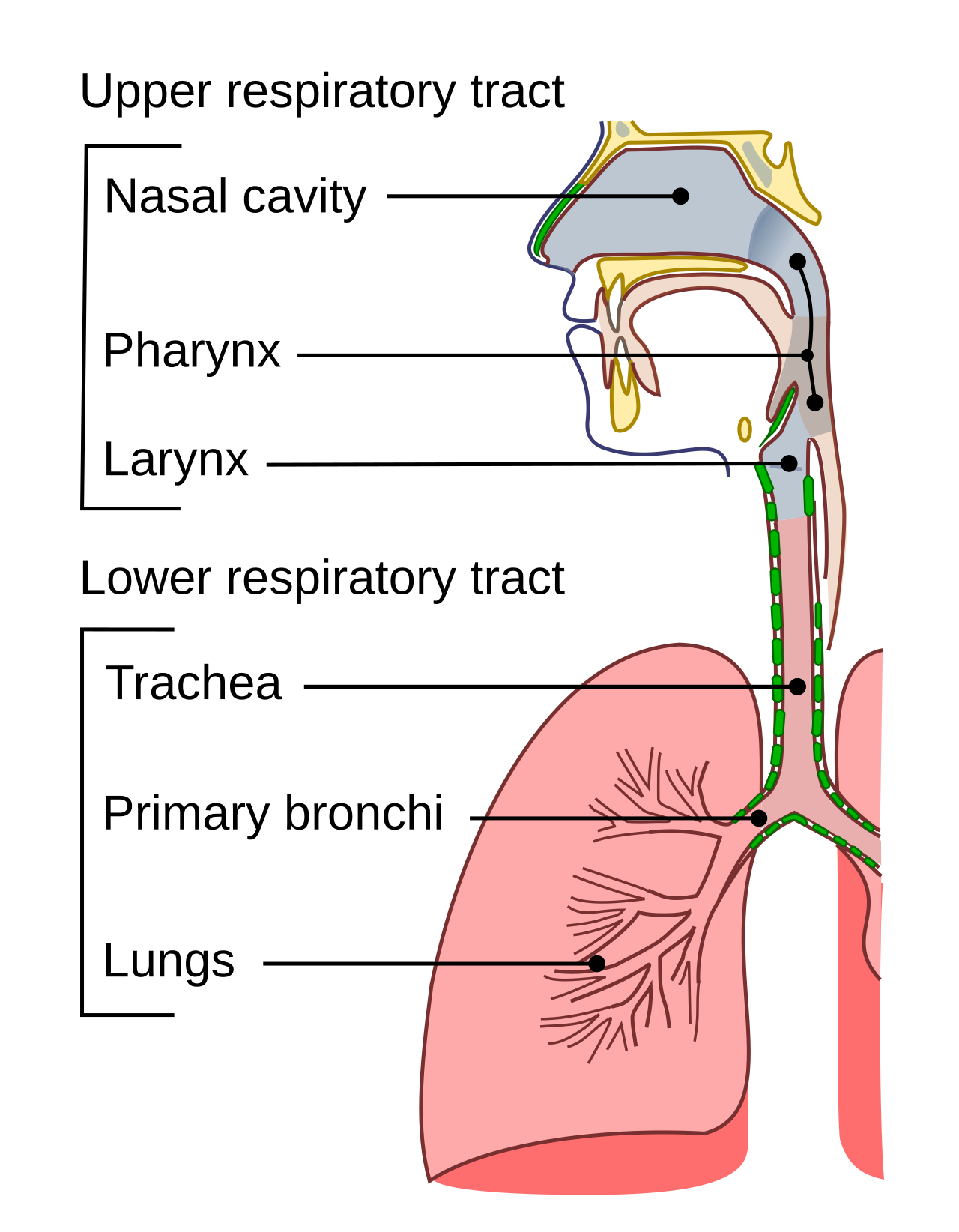
Figure 1 Respiratory Tract
Adequate intake of nutrients is essential for maintaining immune resilience, particularly in their role in supporting the body’s defense against infections, inflammation, and respiratory health. Vitamins such as C and D, minerals like zinc and selenium, omega-3 fatty acids, and bioactive compounds found in functional foods are known to enhance immune function and may specifically benefit lung health by reducing inflammation and supporting the integrity of respiratory tissues. Probiotics and prebiotics also contribute to gut health, which is closely linked to overall immune system regulation, including its impact on respiratory function. 1
The study of nutritional immunology focuses on how these nutrients, supplements, and dietary patterns influence the immune system and contribute to overall health, particularly in inflammation-mediated conditions affecting the lungs. Evidence from animal and human studies highlights the potential of certain nutrients in managing chronic respiratory inflammation, mitigating the effects of infections, and supporting lung tissue repair. These insights provide a foundation for developing dietary strategies to optimize immunity and respiratory health. 2
Significance of the Topic
Resilience against infections refers to the ability to endure and recover from the harmful effects of pathogens. It involves a combination of physiological, genetic, and behavioral factors that help individuals maintain health even when exposed to infectious agents. A strong immune system is the cornerstone of this resilience and plays a vital role in maintaining overall health. Acting as the body’s primary defense, the immune system protects against harmful pathogens, reduces the severity of infections, and speeds up recovery. Its role extends beyond fighting infections, as it also prevents chronic inflammation, which is linked to conditions like diabetes, cardiovascular disease, and neurodegenerative disorders. 3
Infectious diseases are one of the biggest health challenges in the US and each year about 23 million Americans visit a doctor’s office or a clinic seeking treatment for infections. As of 2019, 17.2 billion upper respiratory infections accounted for 42.8% of all-cause illnesses globally. 4 Acute respiratory tract infections are a major cause of global morbidity and mortality and are responsible for 10% of ambulatory and emergency department visits in the USA.5 These infections can also lead to more severe conditions, such as lower respiratory infections, which have higher mortality rates.
The immune system is comprised of the innate and adaptive responses. The innate immune response is rapid and non-antigen specific. It includes physical barriers that prevent pathogen entry, such as the skin and gut epithelium, as well as antimicrobial peptides, the complement system, and various phagocytic and other cells like neutrophils, macrophages, and natural killer cells. These cells detect pathogens using nonspecific pattern-recognition receptors. The innate system acts swiftly to identify and eliminate “non-self” threats, often through inflammatory processes, and subsequently resolves inflammation and repairs any associated damage. However, innate immunity does not improve in speed or effectiveness with repeated exposure to the same pathogen (Figure 2).
Following the innate response, the adaptive immune response is activated. This response involves B lymphocytes (B cells) which can produce antibodies specific to the pathogen, and antigen-specific cells, such as T lymphocytes, which either coordinate the adaptive immune response or kill virus-infected cells. Although slower to activate than the innate system, the adaptive immune system generates immunological “memory,” enabling a robust antigen-specific response to subsequent infections with the same pathogen. This memory forms the basis for how vaccines provide protection against future pathogen exposures.6
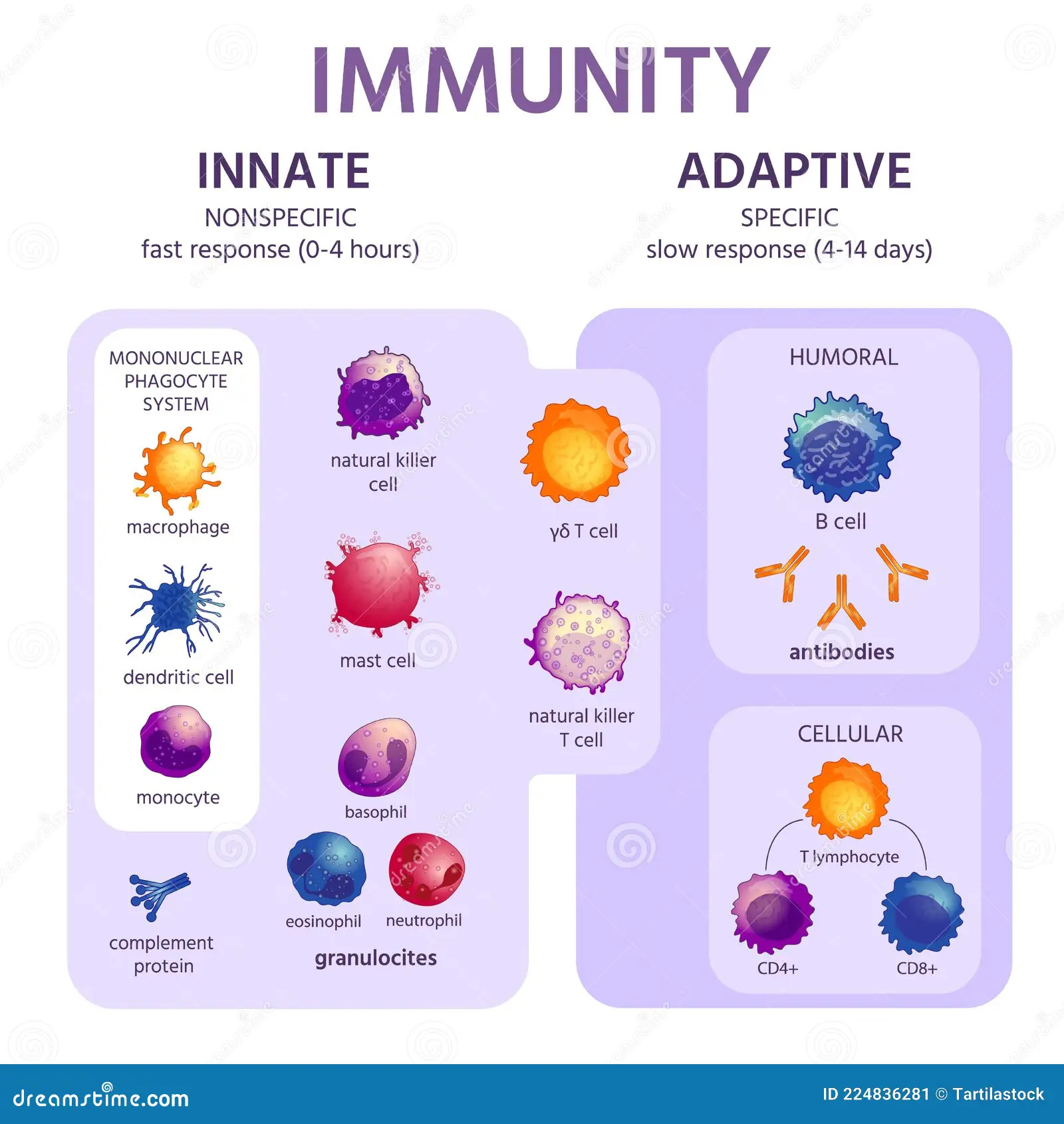
Figure 2 The Innate and Adaptive Immune Response
Nutritional strategies to support optimal immune function are often overlooked in public health discussions about immunity and infection, despite the well-established importance of nutrition in immune health. Several vitamins—such as A, C, D, E, and folate—and trace elements like zinc, and selenium play vital and complementary roles in supporting both the innate and adaptive immune systems. Deficiencies or inadequate levels of these micronutrients can impair immune function and reduce resistance to infections. Additionally, nutrients like omega-3 fatty acids support immune function by helping to resolve inflammation.7
Deficiencies, or even suboptimal levels, of these nutrients can impair immune function, depending on which nutrient or nutrients are lacking. This can lead to a decrease in lymphocyte numbers, impaired phagocytosis and microbial killing by innate immune cells, altered cytokine production, reduced antibody responses, and even compromised wound healing. These functional impairments likely contribute to the clinical immune-related effects of nutrient deficiencies. For example, individuals deficient in vitamin C are more vulnerable to severe respiratory infections like pneumonia.8
Specific Nutrient: Vitamin C
What is it
Vitamin C, or ascorbic acid, is a vital nutrient that humans cannot produce naturally. Unlike most mammals, primates lack the enzyme gulonolactone oxidase, which is essential for synthesizing vitamin C from glucose. This inability stems from genetic mutations that render the enzyme inactive. As a result, humans must rely on dietary sources of vitamin C to prevent deficiency. Without sufficient intake, severe deficiency can lead to scurvy, a potentially life-threatening condition. This makes vitamin C an essential dietary component for maintaining health and survival.
Vitamin C is also well-known for its critical role in supporting various physiological processes, including immune and lung health. Its unique properties as an electron donor, a reducing agent and antioxidant make it indispensable for maintaining overall health and resilience against environmental and physiological stresses. This underscores the importance of ensuring adequate dietary intake of vitamin C for optimal health.
How it Works
Antioxidants are molecules that prevent oxidation, a chemical reaction that produces free radicals capable of initiating chain reactions that damage cells. Free radicals and oxidants are generated either as a result of normal metabolic activities or due to environmental factors such as pollution, cigarette smoke, and radiation. Oxidative stress occurs when the generation of free radicals exceeds the capacity of protective mechanisms to counteract them, contributing to the development of chronic and degenerative diseases, including cancer, autoimmune disorders, aging, rheumatoid arthritis, cardiovascular diseases, and neurodegenerative disorders (Figure 3).9,10
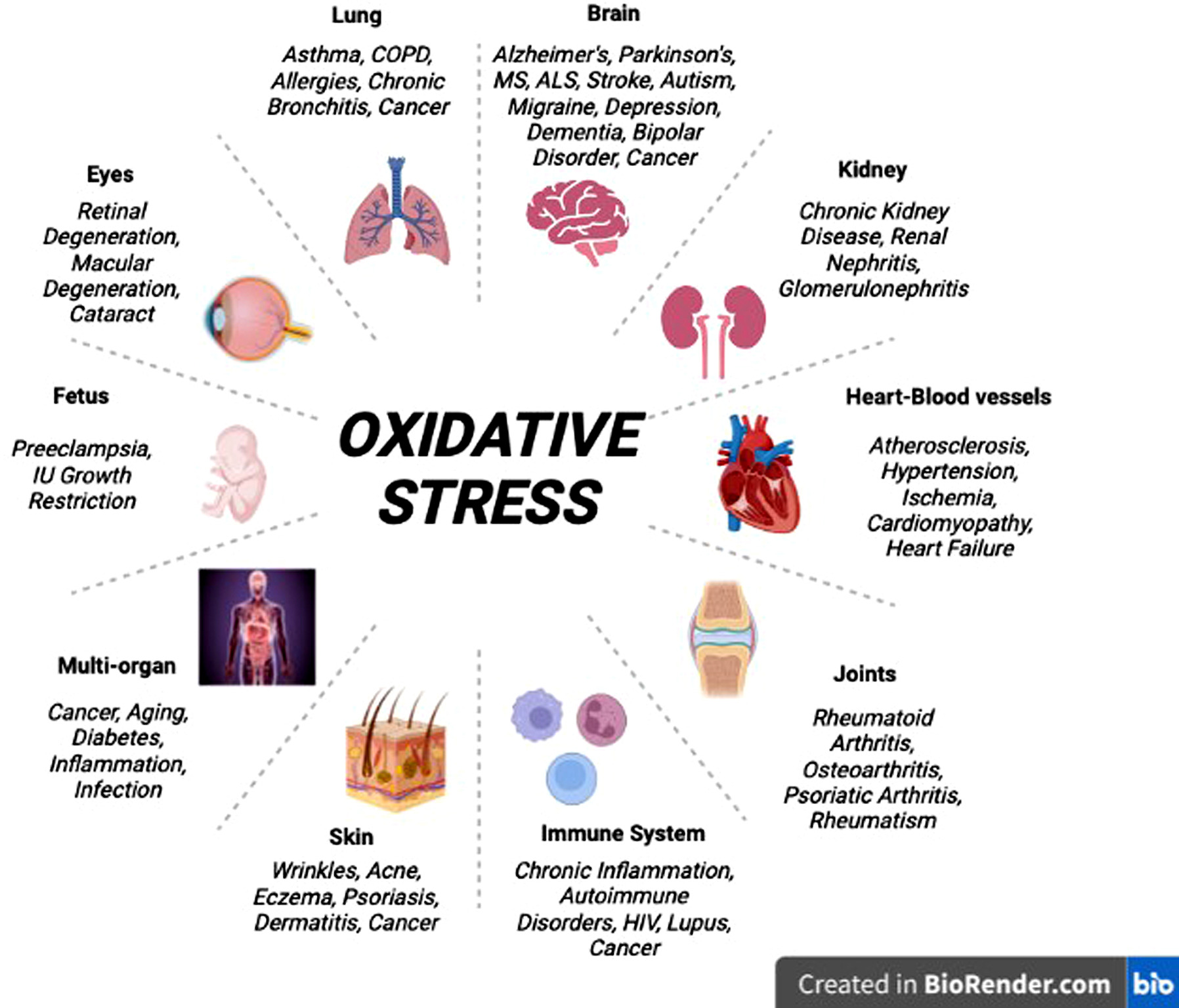
Figure 3 Oxidative stress induced in humans 11
Vitamin C supports the immune system by acting as a powerful antioxidant and serving as a cofactor for essential enzymes. As a water-soluble antioxidant, it works in the cell’s fluid environment to neutralize harmful molecules like oxygen radicals and hydrogen peroxide. Its structure allows it to donate protons and electrons, converting into dehydroascorbic acid (DHA), which can still function biologically. The stable ascorbic radical formed during this process is non-reactive and can be recycled back into vitamin C with the help of specific enzymes, maintaining its protective role in the body.12
In addition, vitamin C contributes to both innate and adaptive immune responses by strengthening epithelial barriers, supporting cellular functions, and modulating cytokine production. By doing so, it helps the body combat infections while also minimizing tissue damage caused by excessive inflammation. 13 Vitamin C significantly enhances the function of neutrophils, the first line of defense in the innate immune response. It accumulates in neutrophils, enhancing their ability to migrate to infection sites (chemotaxis), engulf and kill microbes (phagocytosis), and generate reactive oxygen species for microbial killing. Vitamin C protects neutrophils from oxidative damage during these processes. This helps resolve inflammation and prevents chronic tissue damage.14
In addition to its role in innate immunity, vitamin C enhances adaptive immune responses by supporting the differentiation and proliferation of B- and T-cells. This boosts antibody production and improves the immune system’s ability to target pathogens. Emerging evidence suggests that vitamin C regulates T-cell maturation through epigenetic mechanisms, highlighting its importance in adaptive immunity.15 Vitamin C also plays a significant role in modulating inflammatory responses. It regulates cytokine production, balancing pro-inflammatory and anti-inflammatory signals to ensure a measured immune response. Vitamin C has been shown to reduce histamine levels, which alleviates allergic and inflammatory symptoms. Furthermore, its antioxidant properties protect cells from oxidative damage, which is critical during infections and exposure to environmental pollutants.
Vitamin C deficiency impairs immune function and increases susceptibility to infections, particularly respiratory conditions like pneumonia. Infections further deplete vitamin C levels due to heightened metabolic demands and inflammation.16
What the Research Is Telling Us
Several studies explored the effect of vitamin C in treating infections. A 2013 Cochrane systematic review and meta-analysis examined the effects of vitamin C on common cold incidence, duration, and severity across 29 trials with 11,306 participants. While Vitamin C did not significantly reduce cold incidence, regular supplementation reduced cold duration. Regular supplementation reduced cold duration by 8% in adults and 14% in children, with higher doses (1–2 g/day) shortening colds by 18% in children. Cold severity was also reduced. In contrast, therapeutic use of vitamin C after symptom onset (3,249 episodes) showed no consistent benefit.17
A 2023 study examined the impact of vitamin C on the severity and duration of common cold symptoms through a meta-analysis of 15 comparisons from 10 placebo-controlled trials, involving over 2,700 participants. The trials included oral doses of at least 1 g/day of vitamin C and assessed both mild and severe symptoms using severity scales and proxies such as absenteeism or days confined to the house. The results indicated that vitamin C reduced the severity of colds by an average of 15% (confidence interval 9–21%) and had a more pronounced effect on severe symptoms, shortening their duration by 26% across five trials. In contrast, it showed no significant effect on mild symptoms or the overall cold duration. The analysis highlighted the importance of vitamin C for mitigating severe outcomes, such as reducing days off work or school, especially among high-risk groups like military recruits and athletes. The study suggested the potential of vitamin C for addressing cold-induced complications, including asthma and pneumonia.18
A number of studies have shown that vitamin C supplementation can significantly benefit individuals under physical stress.19 In five trials, vitamin C reduced the incidence of colds by 50% in such individuals, and additional studies demonstrated its effectiveness in alleviating exercise-induced bronchoconstriction.20 These findings highlight the potential of vitamin C as a valuable supplement for physically active individuals who experience respiratory symptoms associated with exercise. The antioxidant properties of vitamin C likely play a key role in mitigating the oxidative stress caused by intense physical activity, making it a promising intervention for improving respiratory health in these populations.
A more recent systematic review and meta-analysis assessed the efficacy and safety of vitamin C in reducing mortality, length of hospital stay (LOS), and other critical outcomes in adults with Community-acquired pneumonia. The analysis included six randomized controlled trials (RCTs) involving 366 patients, and the findings revealed a trend toward reduced mortality in patients receiving vitamin C, although this result was not statistically significant. In terms of hospital length of stay, vitamin C supplementation showed a trend toward a reduction, but this difference was also not statistically significant across all studies. However, when an outlier study was excluded, a significant decrease in hospital length of stay was observed. Further secondary outcomes, such as the duration of vasopressor use, showed promising results. One study reported a significant reduction in the duration of vasopressor support for patients receiving vitamin C. However, other secondary measures, including 30-day readmission risk and time to clinical stabilization, did not show significant benefits from vitamin C supplementation.21
Recent studies have also looked at the effect of vitamin C supplementation in treating COVID-19. A systematic review and meta-analysis analyzed 19 studies. These studies, including 10 randomized controlled trials (RCTs) and 9 retrospective trials, analyzed data from 1,989 COVID-19 patients to assess the effects of vitamin C on in-hospital mortality, ICU length of stay, and adverse events. The findings revealed that vitamin C supplementation reduced in-hospital mortality from 33.9% to 24.1%. In RCTs, the effect was more pronounced, with mortality decreasing from 35.8% to 23.9%. Interestingly, patients treated with vitamin C experienced a longer ICU stay, a finding possibly attributable to differences in disease severity or ICU admission criteria. The analysis highlighted the effectiveness of low doses of vitamin C in reducing mortality, whereas high doses offered no additional benefit and might increase risks.
While the findings on the effect of Vitamin C in treating COVID-19 are promising, the study’s limitations must be noted. There was significant heterogeneity among the included studies in terms of design, patient populations, and treatment protocols. Additionally, the evolving COVID-19 treatment guidelines during the pandemic may have influenced outcomes. Some studies also assessed vitamin C in combination with other supplements, such as zinc and vitamin D, making it challenging to isolate its specific effects. 22
A review examined vitamin C supplementation in the prevention and treatment of pneumonia.23 The review included five studies with a total of 2,655 participants, two of which were randomized controlled trials (RCTs) and three quasi-RCTs. The participants included children under five years, school-aged children, and adults. Two studies focused on vitamin C supplementation for pneumonia prevention, while three evaluated its role as an adjunct to pneumonia treatment. The doses for prevention were 1g-2g daily for 8-14 weeks, and for treatment, 125mg-200mg daily until symptoms resolved. The results showed uncertainty about vitamin C’s effect on pneumonia incidence, and on the duration of illness and hospitalization for treatment. The authors concluded that due to the low number of studies and very low evidence certainty, further high-quality research is needed to assess the role of vitamin C supplementation in pneumonia prevention and treatment.
Another systematic review and meta-analysis evaluated the efficacy and safety of intravenous (IV) vitamin C as a treatment for sepsis, a critical condition caused by a dysregulated immune response to infection. Sepsis is a major cause of mortality and morbidity worldwide, affecting approximately 50 million people annually and contributing to about 20% of all-cause deaths. This meta-analysis systematically reviewed 24 randomized controlled trials (RCTs) involving 3,759 patients. Participants were divided into treatment groups receiving IV vitamin C alone or in combination with other therapies (e.g., thiamine and corticosteroids) and control groups receiving standard care or placebos. The study assessed primary outcomes, such as overall and short-term mortality, and secondary outcomes, including organ dysfunction, adverse events, and duration of hospital stays. The results showed that IV vitamin C appeared to reduce overall mortality, but these reductions were not statistically significant. Similarly, while vitamin C improved Sequential Organ Failure Assessment (SOFA) scores over 72 hours, other outcomes such as hospital mortality, ICU mortality, and the incidence of acute kidney injury (AKI) showed minimal or no improvement. The findings highlighted the variability in the impact of IV vitamin C on sepsis outcomes, attributed partly to differences in study protocols, patient populations, and dosing regimens. Subgroup analyses indicated that the efficacy of vitamin C might be more pronounced in certain conditions, such as monotherapy compared to combination therapies. 24
Critically ill patients often suffer from systemic inflammation, oxidative stress, and multiple organ dysfunction, contributing to increased mortality and prolonged hospital stays. Vitamin C, with its potent antioxidant and anti-inflammatory properties, has been proposed as a therapeutic intervention. A meta-analysis of randomized controlled trials investigated the efficacy of vitamin C in critically ill patients based on high-quality randomized controlled trials (RCTs).25 The study included 18 RCTs comprising 2,001 patients, with 1,005 receiving IV vitamin C and 996 in control groups. It assessed outcomes such as intensive care unit (ICU) and hospital length of stay (LOS), mortality rates, organ dysfunction as measured by Sequential Organ Failure Assessment (SOFA) scores, and adverse events. Results indicated that IV vitamin C reduced ICU LOS by an average of 0.36 days and hospital LOS by 1.5 days. However, it did not significantly affect mortality rates at the longest follow-up, hospital mortality, or ICU mortality. Similarly, there was no statistically significant improvement in SOFA scores. The findings highlighted the potential of IV vitamin C in shortening ICU and hospital stays, especially when administered in high doses (≥3 g/day). However, these reductions, while statistically significant, were clinically modest. Furthermore, the effects on mortality and organ dysfunction were inconclusive, with trial sequential analyses (TSAs) failing to confirm definitive benefits. The heterogeneity among studies, varying patient conditions, and differences in dosing regimens limit the generalizability of the results. 26
Dosage Recommendation Based on Research
The recommended dietary allowances for vitamin C are based on its known physiological and antioxidant functions in white blood cells and are much higher than the amount required for protection from deficiency. The recommended dietary allowance for vitamin C, according to the Food and Nutrition Board (FNB) at the Institute of Medicine (IOM) of the National Academies (formerly National Academy of Sciences), is 90 mg per day for men and 75 mg for women. This dietary allowance is higher for pregnant, and nursing women and for individuals who smoke. High doses of vitamin C (6-8 g/day) may further shorten viral upper respiratory infections by approximately 20%. 27 However, such doses are typically recommended for short-term use and under medical supervision due to the risk of side effects like gastrointestinal discomfort.
Table 1 Dosage Recommendation for XXX
| What’s the Daily Recommended Intake? | How much do I take? | Don’t take more than… | When will I see results? | |
|---|---|---|---|---|
| Vitamin C | 75 mg for women, 90 mg for men | 1-2 g a day | 2000 mg | 1-2 weeks |
Facts and Misconceptions
There are some misconceptions surrounding the effectiveness of vitamin C. One of the common myths is that vitamin C cures the common cold. As discussed above, vitamin C boosts the immune system, may reduce the severity of common cold and shorten its duration, butit does not cure common cold.
There is no evidence that vitamin C cures COVID-19, either. The study described in this review suggests it may reduce in-hospital mortality and offers some benefits as an adjunctive treatment, but it is not a cure.
FDA does not require food labels to list vitamin C content unless it has been added to the food. Foods providing 20% or more of the daily value (DV) are considered to be high sources of a nutrient, but foods providing lower percentages of the DV also contribute to a healthful diet.
Food Sources
Unlike many other vitamins, vitamin C is generally present in relatively high amounts in various foods, ranging from 10–100 mg per 100 g, and in some cases, reaching gram levels per 100 g of fresh weight. This abundance may be attributed to vitamin C being synthesized from sugars, which are widely distributed among different organisms. To date, the synthesis of vitamin C has been observed in all plant species, including algae and photosynthetic protists.
A significant portion of daily vitamin C intake for most individuals comes from regularly consuming fruits and fruit juices, with the specific types of fruits varying by region. Globally, excellent sources of vitamin C include star fruit, guava, black currant, kiwi, and strawberry. While citrus fruits contain less vitamin C compared to the richest sources, they still provide a sufficient amount. Among vegetables, cruciferous varieties such as broccoli, kale, and peppers are particularly rich in vitamin C.
Red Pepper (raw, sweet): 95 mg per ½ cup
Orange juice: 93 mg per ¾ cup
Orange: 70 mg per a medium fruit
Grapefruit juice: 70 mg per ¾ cup
Kiwifruit: 64 mg per a medium fruit
Green pepper (raw, sweet): 60 mg per ½ cup
Broccoli (cooked): 51 mg per ½ cup
Strawberries (fresh, sliced): 49 mg per ½ cup
Brussels sprouts (cooked): 48 mg per ½ cup
Grapefruit: 39 mg per a medium fruit
Broccoli (raw): 39 mg per cup
Tomato juice: 33 mg per ¾ cup
Cantaloupe: 29 mg per ½ cup
Cabbage (cooked): 28 mg per ½ cup
Cauliflower (raw): 26 mg per ½ cup
Potato (baked): 17 mg per a medium potatoe
Tomato (raw): 17 mg per a medium fruit
Spinach (cooked): 9 mg per ½ cup
Green peas (frozen, cooked): 8 mg per ½ cup
Supplemental Sources
Supplements typically contain vitamin C in the form of ascorbic acid, which has the same bioavailability to that of naturally occurring ascorbic acid in foods, such as orange juice and broccoli. 28 While synthetic and food-derived vitamin C are equally bioavailable, consuming vitamin C as part of whole foods is generally preferred. This approach provides additional health benefits through the intake of various macro- and micronutrients, along with phytochemicals present in the food. Epidemiological studies have consistently shown that higher consumption of fruits and vegetables is linked to a reduced risk of stroke, coronary heart disease, and several types of cancer. Vitamin C levels in the body are one of the best indicators of fruit and vegetable consumption, and food-derived vitamin C is associated with a lower incidence of many chronic diseases. However, it remains unclear whether these health benefits are directly attributable to vitamin C or other plant-derived compounds. 29
Side Effects
High oral doses of vitamin C, typically between 5-10 grams taken at once, can result in transient gastrointestinal disturbances, such as osmotic diarrhea and abdominal bloating or pain. These side effects are self-limiting and generally not harmful but may cause discomfort. For most individuals, doses exceeding 1 gram per day are not recommended, as they do not confer significant additional health benefits and may increase the risk of adverse effects. 30 In intravenous administration, even very high doses of vitamin C, ranging from 1 to 200 grams, are usually well tolerated. A study examined the use of intravenous (IV) vitamin C by Complementary and Alternative Medicine (CAM) practitioners, highlighting its widespread application, particularly for infections, cancer, and fatigue. Over the course of 2 years, over 20,000 patients were treated, with an average dose of 28 grams every four days across 22 sessions per patient. The study found that side effects were uncommon and typically minor, such as lethargy, vein irritation, or changes in mental status. However, serious adverse effects, including renal failure and hemolysis, occurred in patients with pre-existing conditions like renal impairment or glucose-6-phosphate dehydrogenase deficiency.31
Legal Status and Availability
Vitamin C is legally recognized worldwide as an essential nutrient required for human health, as the body cannot produce it and its deficiency leads to scurvy. Health authorities, such as the World Health Organization (WHO) and the U.S. Food and Drug Administration (FDA), include it in dietary guidelines, with Recommended Dietary Allowances (RDAs) established based on age and life stage. It is commonly regulated as a dietary supplement under frameworks like the U.S. Dietary Supplement Health and Education Act (DSHEA), which allows its sale without pre-approval as long as labeling and safety standards are met. Similarly, in the European Union, vitamin C is regulated under the Food Supplements Directive, with member states ensuring compliance. Globally, vitamin C is recognized as safe, with regulations focusing on quality, accurate labeling, and the prohibition of unsubstantiated medical claims.
Vitamin C is also widely used in fortified foods and beverages, subject to food safety standards, and in pharmaceutical applications, such as high-dose treatments for scurvy, which require stricter drug regulation. International organizations like Codex Alimentarius provide guidelines on its use, shaping national regulations globally. Its legal status encompasses its roles in supplements, fortified foods, and pharmaceuticals, reflecting its critical importance to human health.
Bottom Line
Vitamin C is a vital micronutrient with antioxidant properties that supports various cellular functions and enhances the immune system through mechanisms such as promoting the differentiation and proliferation of B- and T-cells. It is also critical for the production of interferon, an antiviral cytokine essential in the early stages of viral infections, including influenza. Regular vitamin C supplementation has been shown to reduce the duration of colds in adults and children, although it does not significantly lower their incidence. However, in physically active individuals, vitamin C supplementation can halve the number of colds, and in children, it has been observed to shorten the duration of upper respiratory tract infections.
The benefits of vitamin C extend to severe infections, including pneumonia and COVID-19. High doses, particularly when administered intravenously, may reduce the severity and duration of these infections, with evidence linking intravenous vitamin C to reduced mortality and shorter hospital stays in severe respiratory cases. Notably, high doses of vitamin C are well tolerated. Despite its promise, the effectiveness of vitamin C supplementation can vary depending on dosage and population characteristics. While high doses may be necessary for treating established infections, further research is needed to clarify its role across different infection types and patient groups. Given its safety, affordability, and potential benefits, vitamin C is an appealing candidate for further investigation, particularly in high-risk populations and severe cases.
Specific Nutrient: Vitamin D
What Is It
Vitamin D, also known as calciferol, is a group of fat-soluble secosteroids essential for calcium and phosphate homeostasis in the human body. The main forms of Vitamin D are vitamin D2 (ergocalciferol) and vitamin D2 (cholecalciferol). Vitamin D2 is human-made and added to foods, while vitamin D3 is synthesized in the skin through UVB exposure and obtained from animal-based foods. Vitamin D3 production depends on UVB exposure. Human skin is the main site in which D3 is synthetized by UVB mediated chemical modification of 7- dehydrocholesterol. Vitamin D levels are affected by 7-dehydrocholesterol availability, and factors like season, latitude, clothing, and age. Vitamin D2 and D3 are equally efficiently metabolized by the liver to generate 25-hydroxy vitamin D (calcidiol) by the cytochrome P450 enzyme CYP2R1. 25-hydroxy vitamin D binds to vitamin D binding protein and undergoes further hydroxylation in the liver and kidneys to create the active form of vitamin D, 1,25 dihydroxy vitamin D (Calcitriol). A deficiency in vitamin D impairs calcium absorption, leading to conditions such as rickets in children and osteomalacia in adults, which are characterized by weakened and deformed bones. Severe deficiencies may also contribute to osteoporosis and other metabolic bone diseases.
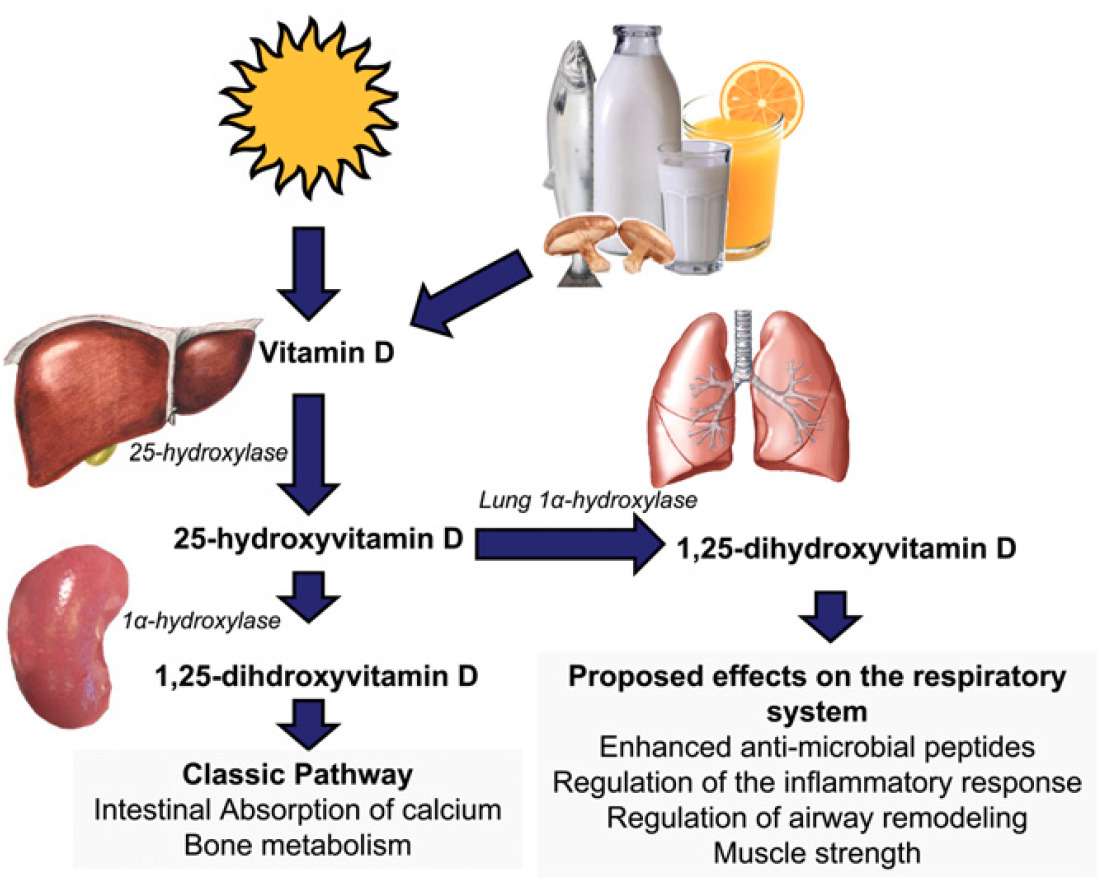
Figure 4 How Vitamin D is Produced
How It Works
Calcitriol, the active form of vitamin D, exerts its multiple biological activities through the binding to the nuclear ligand binding domain (LBD) of the VitD Receptor (nVDR) that functions as a transcription factor. 32 The complex calcitriol-VDR regulates the expression of thousands different genes involved in the regulation of differentiation, activation and proliferation of many cell types, including immune and inflammatory cells :
Regulating Cytokine Production: Calcitriol decreases pro-inflammatory cytokines such as IL-12, IFN-γ, Tumor Necrosis Factor-α, IL-6, IL-8, IL-17, while increasing anti-inflammatory cytokines such as IL-4, and IL-5 via NF-κB inhibition.
Dendritic Cells: Reduces their ability to activate T cells by decreasing co-stimulatory molecules (CD40, CD80, CD86). These dendritic cells are poor inducers of T cell proliferation and activation, induce the differentiation of Treg cells and activate apoptosis.
Macrophages & Monocytes: Calcitriol shifts macrophages toward an anti-inflammatory profile, suppressing COX-2 and NF-κB signaling, and enhances antimicrobial activity via cathelicidin production.
B Cells: Inhibits proliferation, antibody production, and plasma cell differentiation while upregulating IL-10, reducing T cell stimulation.
Airway Epithelial Cells & Antimicrobial Defense: Human airway epithelial cells express both VDR and 1α-hydroxylase, allowing them to locally produce calcitriol. This stimulates the production of antimicrobial peptides like cathelicidin and defensin β4, which help combat bacterial and viral infections. Cathelicidin also exhibits antiviral properties against influenza and respiratory syncytial virus (RSV). Additionally, calcitriol upregulates the innate immune protein hCAP-18, which enhances the body’s ability to kill viruses and bacteria.
Calcitriol plays a significant role in mitigating asthma, a chronic inflammatory condition characterized by airway obstruction and inflammation. Its effects on various cell types contribute to reduced airway hyper-responsiveness, inflammation, and remodeling. In airway muscle cells, calcitriol decreases cell proliferation, mucus secretion, and the production of inflammatory cytokines and matrix metalloproteinases. Within innate immunity, it inhibits differentiation, migration, and cytokine production from mast cells, neutrophils, and eosinophils, which are key contributors to asthma-related inflammation. In children, low serum 25-hydroxy vitamin D levels correlate with a higher risk of asthma, more frequent exacerbations, and reduced lung function, although some studies suggest no direct connection between vitamin D levels and airway inflammation or allergies. In adults, vitamin D insufficiency has been associated with severe or uncontrolled asthma and a greater decline in lung function, particularly in non-smokers and those not using inhaled corticosteroids. Elderly asthma patients are frequently vitamin D deficient, and deficiency in adults has been linked to increased all-cause mortality. While calcitriol appears to regulate immune responses and reduce inflammation in asthma, its clinical role in prevention and treatment remains a subject of ongoing research.33
What the Research Is Telling Us
Observational studies report consistent independent associations between low serum concentrations of 25-hydroxyvitamin D (the major circulating vitamin D metabolite) and susceptibility to acute respiratory tract infection. 34 Various epidemiological and in-vitro data suggested mechanisms by which vitamin D protects against respiratory infections. These have prompted numerous randomised controlled trials to determine whether vitamin D supplementation can decrease the risk of acute respiratory tract infection. A systematic review and meta-analysis assessed the efficacy of vitamin D supplementation in reducing the risk of such infections through data from 25 randomized controlled trials involving 11,321 participants aged 0–95 years across 15 countries. 35 Results indicate that supplementation reduces the risk of acute respiratory tract infections overall, with stronger protective effects in participants with severe vitamin D deficiency (baseline levels <25 nmol/L) and those receiving daily or weekly doses without the administration of a single large dose. Subgroup analyses highlight variations based on dosing frequency, baseline vitamin D levels, and participant characteristics. The study concluded that vitamin D supplementation is safe and most beneficial for those with profound deficiencies, supporting public health measures like food fortification.36
A 2021 study presented an updated meta-analysis examining whether vitamin D supplementation reduces the risk of acute respiratory infections. 37 This study built on the previous 2017 meta-analysis38 by incorporating more recent RCTs to assess the overall effect and identify potential influencing factors. The research team systematically reviewed data from 46 RCTs involving over 75,000 participants. They analyzed aggregated study-level data based on factors such as baseline vitamin D levels, dosage, frequency of supplementation, trial duration, and participant age. The primary outcome was the proportion of participants who experienced at least one acute respiratory infection, and statistical methods were used to compare those receiving vitamin D supplementation with those in placebo or low-dose vitamin D control groups.
The results showed a modest overall reduction in acute respiratory infection risk among participants taking vitamin D supplements compared to those receiving placebos. However, the most significant protective effects were observed in trials where vitamin D was administered daily at doses between 400–1000 IU for up to 12 months and among children aged 1–15 years. No significant protective effect was found when vitamin D was given in single high doses or for participants with higher baseline vitamin D levels. Additionally, the study found no increased risk of serious adverse effects related to supplementation. Despite some observed heterogeneity across trials, the study concluded that vitamin D supplementation is a safe intervention with a small but beneficial effect in reducing the risk of acute respiratory infections, particularly when administered daily at moderate doses. 39
The potential of high-dose vitamin D supplementation in improving clinical outcomes for COVID-19 patients was investigated. A systematic review and meta-analysis of 13 randomized controlled trials evaluated vitamin D’s impact on the length of hospital stay, intensive care unit admissions, and mortality rates. 40 The RCTs focused on patients diagnosed with COVID-19 who received vitamin D supplementation during treatment. The Cochrane Risk of Bias tool was employed to assess study quality, revealing variability in the rigor of included trials. Statistical analysis using a random-effects model accounted for differences across studies.The analysis showed mixed results. Vitamin D supplementation was associated with a reduction in ICU admissions. However, it did not reduce the length of hospital stay or mortality. The authors recommended large-scale, standardized RCTs to better understand the role of vitamin D in managing COVID-19.
The evidence on vitamin D supplementation for preventing respiratory tract infections in children is mixed. Some studies suggest that high-dose vitamin D supplementation does not significantly reduce the incidence of upper respiratory tract infections but lower the incidence of influenza, colds, coughs, and fevers in children. 41 Conversely, vitamin D deficiency has been associated with an increased risk of acute lower respiratory infections in children, indicating that maintaining adequate vitamin D levels might be beneficial. 42 In addition, observational studies suggest supplementation with vitamin D may help reduce the frequency of respiratory infections and asthma exacerbations in children and individuals with chronic respiratory conditions such as asthma. 43
Dosage Recommendations Based on Research
The recommended dietary allowances for vitamin D are established by an established committee of the Food and Nutrition Board (FNB) to indicate daily intakes sufficient to maintain bone health and normal calcium metabolism in healthy people. Even though sunlight is a major source of vitamin D for some people, the FNB based on the vitamin D recommended dietary allowance on the assumption that people receive minimal sun exposure.
| What’s the Daily Recommended Intake? | How much do I take? | Don’t take more than… | When will I see results? | |
|---|---|---|---|---|
| Vitamin D | 15 mcg (600 IU) for women, 15 mcg (600 IU) for men | 10-25 mcg (400-1000 IU) for 12 months or less | 100 mcg (4000 IU) | 4-12 weeks. Best results when up to 12 months. |
Mcg: microgram. IU: International Units. 1 mcg vitamin D is equal to 40 IU.
Facts vs Misconceptions
Vitamin D has been surrounded by myths regarding its role in COVID-19. Contrary to popular belief, there is no direct evidence that vitamin D can prevent or cure COVID-19. Additionally, taking high doses of vitamin D is not always safe and can lead to toxicity, such as hypercalcemia. Evidence shows that vitamin D plays a crucial role in supporting immune health by regulating immune responses and reducing inflammation, which may help manage severe respiratory conditions. Low vitamin D levels are linked to increased susceptibility to severe COVID-19 outcomes, particularly in high-risk groups like the elderly and obese individuals. While some clinical trials suggest potential benefits of vitamin D in COVID-19 patients, there is no consensus on its ability to reduce mortality or severe outcomes. Overall, vitamin D supports general health but is not a proven treatment or preventative measure for COVID-19. 44
Food Sources
Cod liver oil: 34.0 mcg per 1 tablespoon
Trout (rainbow), farmed, cooked: 16.2 mcg per 3 ounces
Salmon (sockeye), cooked: 14.2 mcg per 3 ounces
Mushrooms, white, raw, sliced, exposed to UV light: 9.2 mcg per ½ cup
Milk, 2% milkfat, vitamin D fortified: 2.9 mcg per 1 cup
Soy, almond, and oat milks, vitamin D fortified, various brands: 2.5–3.6 mcg per 1 cup
Ready-to-eat cereal, fortified with 10% of the DV for vitamin D: 2.0 mcg per 1 serving
Sardines (Atlantic), canned in oil, drained: 1.2 mcg per 2 sardines
Egg, 1 large, scrambled: 1.1 mcg per 1 egg
Liver, beef, braised: 1.0 mcg per 3 ounces
Tuna fish (light), canned in water, drained: 1.0 mcg per 3 ounces
Cheese, cheddar: 0.4 mcg per 1.5 ounces
Supplemental Sources
Side Effects
While vitamin D supplementation is generally safe, long-term use has been associated with increased risk of hypercalcemia (elevated calcium levels in the blood) and hypercalciuria (elevated calcium levels in the urine).45 46 These conditions occur regardless of the vitamin D dose and duration, and are not significantly influenced by baseline vitamin D levels or calcium supplementation. The risk of developing kidney stones from vitamin D supplementation is not significantly increased. In extreme cases, vitamin D toxicity causes renal failure, calcification of soft tissues throughout the body, cardiac arrhythmias and even death. Vitamin D toxicity has been caused by the consumption of dietary supplements that contained excessive vitamin D amounts. 47
Drug interactions: Vitamin D supplements can interact with various medications, potentially affecting their efficacy or causing unintended side effects. For example, the weight-loss drug orlistat (Xenical and alli) can reduce vitamin D absorption, lowering 25(OH)D levels. Statins, which lower cholesterol, might reduce vitamin D synthesis since vitamin D is derived from cholesterol, and high doses of vitamin D may decrease the effectiveness of certain statins, such as atorvastatin and simvastatin, due to competition for metabolizing enzymes. Corticosteroids like prednisone can impair vitamin D metabolism and reduce calcium absorption, with deficiency rates being higher in steroid users. Thiazide diuretics, which decrease calcium excretion, combined with vitamin D supplements, can lead to hypercalcemia, especially in older adults or those with renal issues. 48
Legal Status and Availability
The legal status of vitamin D depends on its classification and intended use. In most cases, vitamin D is categorized as a dietary supplement. In the United States, it is legally available over the counter under the Dietary Supplement Health and Education Act (DSHEA) of 1994 and is commonly sold in forms like vitamin D2 (ergocalciferol) or vitamin D3 (cholecalciferol). In higher doses, vitamin D may require a prescription, as is the case with ergocalciferol at 50,000 IU in the U.S., used to treat conditions like vitamin D deficiency, osteomalacia, or rickets.
Vitamin D is also legally added to certain foods, such as milk, cereals, and orange juice, under food fortification programs designed to prevent deficiencies. These programs and the regulations governing them vary between countries. In some regions, especially in Europe, high-dose vitamin D supplements are classified as medicinal products and may only be sold in pharmacies or with a prescription. Additionally, in a few developing nations, access to vitamin D supplements may be limited due to public health regulations or supply constraints.
Bottom Line
Vitamin D plays a crucial role in maintaining calcium and phosphate balance, supporting immune function, and respiratory health. It exists in two main forms: D2 (ergocalciferol), found in fortified foods and some plants, and D3 (cholecalciferol), which is synthesized in the skin through UVB exposure and is found in animal-based foods. The active form, calcitriol, binds to the vitamin D receptor (VDR), regulating gene expression and influencing various physiological processes, including immune responses and inflammation control.
One of the most well-established benefits of vitamin D is its role immune regulation, assisting in cytokine production and immune cell activity. Some research suggests that adequate vitamin D levels may reduce the risk of acute respiratory infections and support asthma management.
Regarding COVID-19, there is no definitive evidence that vitamin D prevents or cures the disease. However, studies indicate that individuals with sufficient vitamin D levels may have better immune responses and lower risks of severe respiratory complications. While high-dose supplementation does not significantly reduce COVID-19 mortality, moderate daily doses (400-1000 IU) over an extended period may help reduce respiratory infections, particularly in individuals with vitamin D deficiencies. The effects on respiratory infections in children remain mixed, though some studies suggest a lower risk of colds and flu.
The recommended daily intake of vitamin D for adults is 600 IU (15 mcg), with supplementation in the range of 400-1000 IU per day being generally effective. The upper limit is 4000 IU per day, beyond which there is a risk of toxicity. Natural food sources rich in vitamin D include cod liver oil, fatty fish like salmon and trout, UV-exposed mushrooms, fortified milk, eggs, and cheese. While vitamin D is essential for health, excessive intake can lead to hypercalcemia, which may cause kidney damage, cardiovascular issues, and other complications. Certain medications, such as orlistat, statins, and corticosteroids, can also interfere with vitamin D absorption or metabolism.
Specific Nutrient: Zinc
What Is It
Zinc is a trace element and an essential mineral found naturally in the Earth’s crust, water, and biological systems. It is a key component of various enzymes and proteins, making it fundamental to the structure and function of cells. It is also involved in many aspects of cellular metabolism, and plays a role in enhancing immune function. As a dietary mineral, zinc is present in foods like meat, seafood, and nuts and is required in small but consistent amounts to support overall health. The total amount of zinc in the body is approximately 1.5 g in women and 2.5 g in men. Most of the zinc is stored in skeletal muscle and bone.
How It Works
Zinc plays a pivotal role in the immune system, influencing both innate and adaptive immunity. It is essential for the development, activation, and regulation of immune cells, including T cells, B cells, macrophages, and natural killer (NK) cells. Zinc’s involvement begins at the foundational level, supporting hematopoiesis (the formation of blood cells) and the differentiation of immune cell subtypes. During immune responses, zinc is critical for signal transduction pathways that regulate immune cell function. 49 50 For example, it enables T cells to proliferate and perform their roles in identifying and neutralizing pathogens (via Toll-like receptors), while B cells rely on zinc for the production of antibodies necessary to target specific invaders. Macrophages, as part of the innate immune system, depend on zinc for effective phagocytosis, which allows them to engulf and destroy bacteria, viruses, and other harmful particles. 51
Zinc also directly affects the immune system’s ability to combat infections. It enhances NK cell activity, which is vital for the early elimination of virus-infected cells. Additionally, zinc modulates the production of cytokines, small proteins that mediate communication between immune cells and coordinate inflammatory responses. By regulating cytokine levels, zinc helps maintain a balanced immune response, preventing excessive inflammation that can damage healthy tissues. 52
The correlation between zinc deficiency and weakened immunity is well-documented. A lack of zinc impairs the proliferation and function of T cells, reduces antibody production, and compromises the innate immune system’s ability to respond to infections. This deficiency increases susceptibility to respiratory infections, gastrointestinal illnesses, and slower wound healing.53 Conversely, zinc supplementation has been shown to bolster immune defenses, reducing the severity and duration of illnesses like the common cold and enhancing vaccine responses. This underscores the importance of maintaining adequate zinc levels for optimal immune function, especially in at-risk groups such as children, the elderly, and individuals with chronic health conditions. Balancing zinc intake is critical, as both deficiency and excess can disrupt immune regulation, demonstrating zinc’s intricate and indispensable connection to immune health.54 Serum or plasma zinc levels are typically used in clinical practice to assess zing status. According to the NIH, Office of Dietary Supplements, in healthy people, the amount of zinc in serum or plasma is 80 to 120 mcg/dL (12 to 19 mcmol/L).
What the Research Is Telling Us
Clinical trials evaluating the effects of zinc supplements on the common cold have yielded inconsistent results. However, evidence suggests that zinc, taken in lozenge or syrup form soon after the onset of symptoms, can shorten the duration of the common cold, though it does not appear to reduce symptom severity.55 56
A 2021 systematic review and meta-analysis evaluated the role of zinc in preventing and treating acute viral respiratory tract infections (RTIs) in adults. Data were drawn from 28 randomized controlled trials involving 5,446 participants (mostly adults younger than 65 years) who had a community-acquired viral respiratory tract infection or were inoculated with a rhinovirus. The study focused on various zinc delivery methods, including oral, sublingual, and intranasal administration, and assessed their effects on preventing RTIs, reducing symptom duration and severity, and potential adverse events. 57 It was found that zinc supplementation significantly reduced the risk of developing symptoms of viral RTIs by 32% compared to placebo. It was also found that zinc shortened the duration of symptoms by approximately two days. While zinc was generally safe, non-serious adverse effects such as nausea and gastrointestinal discomfort were more frequently reported, with the type of side effects varying depending on the formulation. The findings of this systematic review suggest that zinc may be a safe and accessible option for reducing the incidence and severity of viral RTIs, particularly when used preventatively or as an early treatment. 58 The author of an earlier systematic review concluded that the use of zinc lozenges at doses of over 75 mg/day reduced the duration of the common cold, whereas lower doses did not. 59
Another meta-analysis and systematic search was conducted across multiple databases and trials registries, identifying 34 randomized controlled trials involving 8526 participants. These trials assessed various forms of zinc, with lozenges being the most common method of administration. The results indicated that zinc may have little or no effect on preventing the onset of a cold, with low-certainty evidence showing no reduction in cold risk. In terms of treatment, zinc supplementation likely reduced the duration of colds, though evidence on symptom severity and ongoing cold risk was inconclusive. Additionally, zinc supplementation was associated with an increase in non-serious adverse events, such as an unpleasant taste or stomach cramps.60
Several clinical trials have investigated the impact of zinc supplementation on reducing the incidence of pneumonia and its use as an adjunctive treatment. A 2016 Cochrane Review of six trials conducted in low-income countries evaluated 5,193 children aged 2 to 59 months who received 10–20 mg of zinc daily for up to 20 months. The results showed a lower incidence and prevalence of pneumonia compared to placebo.61
However, most evidence indicates that using zinc supplements as an adjunct to treat pneumonia in children does not significantly impact mortality rates or recovery time. A 2020 systematic review and meta-analysis analyzed 11 trials involving children aged 2 to 60 months, primarily with severe pneumonia, in low- and middle-income countries. 62 The findings revealed no significant differences in mortality rates or recovery times between children receiving 10–20 mg/day of zinc for 7–14 days (or until discharge) and those receiving a placebo.
Although clinical evidence is currently lacking on whether zinc has a role in protecting against COVID-19, initial findings suggest that optimizing zinc status may offer benefits in combating the virus. In vitro studies have demonstrated that Zn²⁺ ions possess antiviral properties, including the ability to inhibit SARS-CoV RNA polymerase activity. Furthermore, indirect evidence indicates that Zn²⁺ might reduce the activity of angiotensin-converting enzyme 2 (ACE2), the receptor for SARS-CoV-2. 63
Researchers found that zinc may enhance antiviral immunity by upregulating interferon-α production and increasing its effectiveness. It also exhibits anti-inflammatory properties by inhibiting NF-κB signaling and regulating T-cell function as mentioned earlier in this review, which may help mitigate the cytokine storm associated with severe COVID-19 cases.
Additionally, zinc supplementation could lower the risk of bacterial co-infections by improving mucociliary clearance, strengthening the respiratory epithelium’s barrier function, and exerting direct antibacterial effects against pathogens such as Streptococcus pneumoniae. Therefore, zinc holds potential as a preventive and adjunctive therapy for COVID-19 by reducing inflammation, enhancing mucociliary clearance, preventing ventilator-induced lung injury, and modulating both antiviral and antibacterial immunity. However, more clinical and experimental studies are needed to validate these promising findings.64
Dosage Recommendation Based on Research
The recommended dietary allowances for zinc according to the Food and Nutrition Board (FNB) at the Institute of Medicine (IOM) of the National Academies (formerly National Academy of Sciences) is 11 mg per day for men and 8 mg for women. This dietary allowance is higher for pregnant, and nursing women.
| What’s the Daily Recommended Intake? | How much do I take? | Don’t take more than… | When will I see results? | |
|---|---|---|---|---|
| Vitamin C | 8 mg for women, 11 mg for men | 10-30 mg per day for children and up to 75 mg for adults | 75 mg per day for adults | 3-4 days |
Phytate, the storage form of phosphorus in plants, is the primary dietary factor that affects zinc absorption. It can bind to zinc in the intestinal lumen, forming an insoluble complex that cannot be digested or absorbed, as humans lack the intestinal phytase enzyme. This inhibitory effect on zinc absorption can be significant in adults, as there is no evidence of an adaptive response to consistently high phytate intake. Due to its impact on zinc absorption, increasing dietary phytate may require the need for a higher dietary zinc requirement. 65
Facts vs Misconceptions
Zinc is often misunderstood, leading to several common misconceptions. Many believe that zinc is a cure-all for illnesses, but it cannot cure or prevent major diseases like pneumonia. Another widespread myth is that zinc can prevent colds entirely, and some even assume zinc supplements are safe at any dose. Despite these misconceptions, zinc is an essential nutrient that plays a role in supporting immune function, wound healing, cell growth, and DNA synthesis. Supplementation with zinc has been shown to reduce the duration of colds.
Food Sources
The richest sources of zinc are meat, fish, and seafood. Among these, oysters provide the highest amount of zinc per serving, but beef accounts for 20% of zinc intake from food in the United States due to its widespread consumption. Eggs and dairy products also supply zinc. Plant-based foods like beans, nuts, and whole grains contain zinc as well, but its bioavailability is lower compared to animal sources because these foods contain phytates.66 Fruits and vegetables contribute minimal amounts of zinc. Fortified breakfast cereals are another significant source of zinc in the U.S. diet.
Oysters (Eastern), farmed, raw: 32 mg per 3 ounces
Oysters (Pacific), cooked: 28.2 mg per 3 ounces
Beef, bottom sirloin, roasted: 3.8 mg per 3 ounces
Blue crab, cooked: 3.2 mg per 3 ounces
Breakfast cereals, fortified with 25% of the DV for zinc: 2.8 mg per 1 serving
Cereals, oats, regular and quick, unenriched, cooked with water: 2.3 mg per 1 cup
Pumpkin seeds, roasted: 2.2 mg per 1 ounce
Pork, center loin (chops), bone in, broiled: 1.9 mg per 3 ounces
Turkey breast, meat only, roasted: 1.5 mg per 3 ounces
Cheese, cheddar: 1.5 mg per 1.5 ounces
Shrimp, cooked: 1.4 mg per 3 ounces
Lentils, boiled: 1.3 mg per ½ cup
Sardines, canned in oil, drained solids with bone: 1.1 mg per 3 ounces
Greek yogurt, plain: 1.0 mg per 6 ounces
Milk, 1% milkfat: 1.0 mg per 1 cup
Peanuts, dry roasted: 0.8 mg per 1 ounce
Rice, brown, long grain, cooked: 0.7 mg per ½ cup
Egg, large: 0.6 mg per 1 egg
Kidney beans, canned: 0.6 mg per ½ cup
Bread, whole wheat: 0.6 mg per 1 slice
Fish, salmon, cooked: 0.5 mg per 3 ounces
Broccoli, chopped, cooked: 0.4 mg per ½ cup
Rice, white, long grain, cooked: 0.3 mg per ½ cup
Bread, white: 0.2 mg per 1 slice
Cherry tomatoes, raw: 0.1 mg per ½ cup
Blueberries, raw: 0.1 mg per ½ cup
Supplemental Sources
Zinc is available in various dietary supplements forms, including standalone zinc supplements, combination products with other ingredients, and multivitamin/mineral formulations. These supplements may contain different forms of zinc, such as zinc sulfate, zinc acetate, or zinc gluconate. The Supplement Facts label specifies the amount of elemental zinc in the product, rather than the weight of the entire zinc-containing compound.
Zinc absorption rates vary depending on the form. For young adults, zinc citrate and zinc gluconate are absorbed at similar rates, approximately 61%, while zinc oxide has a slightly lower absorption rate of 50%.67 Taking zinc supplements alongside supplements containing 25 mg or more of elemental iron can reduce zinc absorption and plasma zinc levels.68 However, the iron added to enriched or fortified foods does not affect zinc absorption.
Side Effects
Zinc toxicity can occur in both acute and chronic forms. Acute toxicity symptoms include nausea, vomiting, loss of appetite, abdominal cramps, diarrhea, and headaches. While doses as low as 50 mg in adults can cause nausea, much higher doses, such as several grams, are associated with severe toxicity. Chronic zinc toxicity has been observed in adults consuming 150–450 mg of zinc daily. Symptoms include gastrointestinal distress, impaired immune function (e.g., reduced lymphocyte response), decreased HDL cholesterol, and low serum copper levels. However, zinc is generally considered a low-toxicity nutrient at supplemental doses below 50 mg/day. 69
The Food and Nutrition Board has set Tolerable Upper Intake Levels (ULs) for dietary zinc to minimize the risk of adverse effects from long-term high intakes. For adults, the UL is 40 mg/day, while ULs for children range from 5 mg/day in infants (6–11 months old) to 34 mg/day in adolescents (14–18 years old).
Zinc-drug interactions are also notable due to zinc’s extensive role in metabolic processes. Therapeutic drugs, including antibiotics, penicillamine, and diuretics, can interact with zinc, altering its function.
Legal Status and Availability
Zinc is available as a dietary supplement in various forms, including single-ingredient zinc supplements, combinations of zinc with other ingredients, and multivitamin/mineral products. These supplements can contain different forms of zinc, such as zinc sulfate, zinc acetate, and zinc gluconate. The Supplement Facts label on dietary supplements specifies the amount of elemental zinc, not the total weight of the zinc compound. Zinc absorption rates vary by form. Supplements with zinc citrate or zinc gluconate have an absorption rate of approximately 61% in young adults, whereas zinc oxide has a lower absorption rate of 50%. 70
Zinc is also found in products like homeopathic remedies and over-the-counter nasal sprays marketed to treat colds, flu symptoms, or support immune health. Some denture adhesive creams contain 17–34 mg/g of zinc to improve adhesion. When used as directed, these products contribute 0.5–1.5 g of zinc per day, which is considered safe. However, excessive and chronic use can cause zinc toxicity, leading to copper deficiency and neurological issues. Cases of toxicity have been reported in individuals using at least one tube of denture cream containing 2.4 g or more of zinc weekly over several years. To address this risk, many denture creams have been reformulated to exclude zinc.71
Bottom Line
Zinc is an essential mineral crucial for enzyme function, immune health, DNA synthesis, wound healing, and growth. Sources include animal-based foods (meat, seafood, dairy), fortified cereals, and some plant-based foods, though absorption is lower from plants due to phytates. When taken in lozenge or syrup form soon after the onset of symptoms, zinc can shorten the duration of the common cold, though it does not appear to reduce symptom severity. Research on the effects of zinc on COVID-19, pneumonia and other respiratory tract infections has been inconclusive.
Specific Nutrient: Elderberry
What Is It
Elderberries are small, dark berries from the Sambucus tree, commonly known as elder. The most common type is Sambucus nigra, or European elderberry. Sambucus nigra is natively spread in European countries and is used as colorant or flavor in juices and wines.

Figure 5 Elderberry
The berries of the elder plant have been traditionally used to address cold and flu symptoms, but elderberry gained renewed attention during the COVID-19 pandemic due to its perceived immune-boosting properties.
How It Works
Elderberry contains anthocyanins, a subclass of flavonoids, which may exhibit immunomodulating and anti-inflammatory properties. 72 These molecules are believed to play a dual role in combating viral infections. Anthocyanins can bind to and neutralize viral glycoproteins, which are essential for viruses to attach to and enter host cells. This may help inhibit viral replication, as shown in Invitro studies where elderberry extracts showed activity against influenza A, influenza B, and H1N1 (“swine flu”) viruses. 73 74
In addition, elderberries appear to influence cytokine production and macrophage activation. 75 Cytokines are signaling proteins that regulate immune responses. Some studies suggest elderberry increases the production of inflammatory cytokines like TNF-alpha and interleukins. 76 This highlights elderberry’s potential benefits to the immune system activation and in the inflammatory process in healthy individuals or in patients with various diseases. to enhance immune defense without overstimulation. Elderberry has also been studied for its ability to reduce inflammation-related markers, such as C-reactive protein (CRP) and interleukins. 77
What the Research Is Telling Us
A randomized, double-blind placebo-controlled clinical trial investigated the effects of elderberry extract on cold symptoms in air travelers. 78 The trial involved 312 participants who took either elderberry extract or a placebo before and during their travels. The results indicated that while the number of cold episodes was similar in both groups, those in the elderberry group experienced significantly shorter cold durations (4.75 vs. 6.88 days) and lower symptom severity. The study concluded that elderberry supplementation could be beneficial in reducing cold severity and duration in travelers.
A meta-analysis of randomized controlled clinical trials found that supplementation with a standardized elderberry extract is significantly effective at reducing the total duration and severity of upper respiratory symptoms, as compared to a placebo group. The authors suggested that elderberry may be a safe alternative to antibiotics and prescription drugs for viral infections like the common cold and influenza. 79A more recent systematic review that assessed research articles and five randomized clinical trials found that elderberry may reduce the duration and severity of cold, and is associated with a lower risk of influenza complications compared to oseltamivir although the evidence is uncertain. 80
During the COVID-19 pandemic, there has been interest in elderberry supplements to treat or prevent illness, but also concern that elderberry might overstimulate the immune system and increase the risk of ‘cytokine storm’.81
Side Effects
Elderberry is generally considered safe for treating vial respiratory illnesses, with no evidence of stimulating the immune system or causing a cytokine storm. However, the evidence on the harms remains uncertain, and further research is needed to confirm these findings.
The barks, leaves, seeds and unripe fruits of elderberry contain a potentially toxic cyanogenic glycoside “sambunigrin”. The amount of cyanogenic glycoside depends on growing conditions and the flowers have lower content of cyanogenic glycoside in comparison with other parts of plant. 82 These cyanide-producing compounds could cause nausea, vomiting and diarrhea.
Facts vs Misconception
While the preliminary research presented above suggests that elderberry may help relieve symptoms of colds, flu, or other upper respiratory infections, the overall body of evidence remains limited. Only a small number of studies have evaluated its effectiveness for these conditions. Very limited research has been conducted on the potential benefits of elderberry for other health purposes. Despite claims made by some companies, the U.S. Food and Drug Administration (FDA) and the Federal Trade Commission have taken action against those marketing elderberry products with unproven claims, particularly regarding COVID-19. Ultimately, there is not enough scientific information to confirm elderberry’s effectiveness for treating illnesses beyond mild respiratory infections, highlighting the gap between popular perception and established medical research.
Legal Status and Availability
Elderberry is available in the U.S. as a dietary supplement, but it is not approved by the FDA as a drug for treating or preventing any disease. Elderberry products are sold in various forms, such as syrups, gummies, capsules, and teas.
As a dietary supplement, elderberry falls under the Dietary Supplement Health and Education Act (DSHEA) of 1994, which allows its sale as long as manufacturers follow FDA regulations regarding labeling, good manufacturing practices, and safety. However, the FDA and the Federal Trade Commission (FTC) have taken action against companies that falsely advertise elderberry products, especially those making unproven claims related to COVID-19.
Elderberry supplements are widely available in health stores, pharmacies, supermarkets, and online retailers. Since they are not regulated as strictly as prescription or over-the-counter medications, consumers should be cautious when purchasing these products and look for reputable brands that provide transparent ingredient information and third-party testing.
Bottom Line
Dietary Strategies for Immune Health
Plant Based Diets
Plant-based diets prioritize minimally processed fruits, vegetables, whole grains, legumes, nuts, seeds, herbs, and spices. It minimizes or excludes animal products while emphasizing nutrient-rich, minimally processed plant foods. A vegetarian diet excludes meat, poultry, and fish but may include dairy, eggs, and other animal-derived products. A vegan diet, on the other hand, eliminates all animal products, including dairy, eggs, and even honey, focusing solely on plant-based foods. These diets are associated with numerous health benefits, including enhanced immune function. Research indicates that healthful plant-based diets play a significant role in promoting respiratory health, reducing risk of respiratory infections,83 and supporting lung function 84. The Academy of Nutrition and Dietetics states that “vegetarian diets, including total vegetarian or vegan diets, are healthful, nutritionally adequate, and may provide health benefits in the prevention and treatment of certain diseases”. These diets are rich in essential macro- and micronutrients, with the notable exceptions of vitamin B12 and, potentially, vitamin D, which are limited or absent in plant-based sources.85 Additionally, plant-based diets offer bioactive compounds, phytochemicals, like saponins, flavonoids, and alkaloids, which have demonstrated immune-boosting effects which may help strengthen the body’s defenses against respiratory infections and improve overall lung health. 86
A 2022 systematic review of 141 studies (2000–2020) evaluated the nutrient profiles of various dietary patterns highlighting key differences that impact immune system health. 87 While plant-based diets, including vegetarian and vegan patterns, provided higher levels of fiber, polyunsaturated fatty acids, folate, vitamins C and E, and magnesium, nutrients known to support immune function by reducing inflammation and oxidative stress, they were associated with lower levels of critical nutrients such as vitamin B12, vitamin D, iodine, calcium, iron (particularly in women), and zinc due to reduced bioavailability. In contrast, meat-inclusive diets offered higher levels of protein and long-chain omega-3 fatty acids (EPA and DHA) but lacked the fiber and antioxidant-rich nutrients found in plant-based diets.
It has been hypothesized that a vegetarian diet rich in flavonoids, carotenoids, and other plant compounds can modulate inflammation and immune function. In addition, the reduced consumption of animal products which typically lead to lower saturated fat intake has been associated with decreased inflammatory markers. A systematic review and meta-analysis of 40 studies found that vegetarian diets were linked to significantly lower levels of C-reactive protein (CRP), fibrinogen, and leukocyte counts—key biomarkers of inflammation—compared to non-vegetarian diets. 88 These findings suggest reduced systemic inflammation and improved immune balance, though high variability among studies limits the robustness of conclusions for some biomarkers.
Plant-based functional foods—natural or processed plant products containing bioactive components—have shown immunomodulatory effects. These foods may offer additional pathways to bolster immune health and mitigate inflammation. For example, garlic contains organosulfur compounds with immunostimulatory properties. It enhances immune responses and detoxification, showing potential in immune support. Green tea is rich in catechins which have antioxidant anti-inflammatory effects. 89
Plant-Based Diets and Disease Outcomes
A 2022 study included 11,956 adults, among whom 2,522 hospitalizations for respiratory infections and 4,308 hospitalizations for any infection were recorded over a median follow-up of approximately 22 years. The study found that higher consumption of plant-based foods was associated with lower incidences of hospitalizations for both respiratory infections and any infection. Specifically, a higher healthy plant-based diet index was linked to a reduced risk of hospitalization for respiratory infections and any infection, while a higher plant-based diet index was associated with a reduced risk of hospitalizations for any infection. 90 This highlights the importance of focusing on the quality of plant-based diets for optimal health outcomes.
A study analyzing the impact of fruit and vegetable intake on COVID-19 outcomes underscores the potential benefits of plant-based diets. Using data from 170 countries, researchers found a negative correlation between vegetable and fruit consumption and COVID-19 incidence and mortality. Key findings included:
In developed countries, low vegetable and fruit intake correlated with higher COVID-19 cases and deaths.
In regions with adequate or high consumption, death rates remained low despite high case numbers.
In developing countries, sufficient intake reduced both cases and deaths.
However, the reliance on machine learning in this study introduces potential biases, such as regional variability in vegetable and fruit intake and COVID-19 prevalence. 91
Mediterranean Diets
The Mediterranean Diet is a nutritional model rooted in the traditional eating habits of countries surrounding the Mediterranean basin, which includes at least 16 nations. While dietary practices differ across countries and even within regions of the same country due to variations in culture, ethnicity, religion, economy, and agricultural production, the Mediterranean dietary pattern (MDP) shares key characteristics.
These include a high intake of olive oil, fruits, vegetables, whole grains, legumes, nuts, and seeds. The MDP also features moderate consumption of fish, shellfish, white meat, eggs, and fermented dairy products like cheese and yogurt, with limited consumption of red meat, processed meats, and sugary foods. Moderate wine consumption, particularly red wine with meals, is also a traditional component.92
The Mediterranean Diet is distinguished by its relatively high fat content, comprising 40%–50% of daily caloric intake, with saturated fatty acids (SFA) making up ≤ 8% and monounsaturated fatty acids (MUFA), primarily from olive oil, accounting for 15%–25%. It includes a high intake of omega-3 fatty acids from fish and plant sources, along with a favorable Omega-6:Omega-3 ratio of 2:1–1:1, compared to 14:1 in Europe. Additionally, the diet is rich in dietary fiber, features a low glycemic index and glycemic load, and provides anti-inflammatory and antioxidant benefits, which collectively contribute to its positive impact on health. 93
Experimental and clinical studies have shown that olive oil down-regulates the expression of several inflammatory biomarkers in circulating lymphocytes and monocytes. 94
Chronic inflammation is a known contributor to various lung conditions, including asthma, chronic obstructive pulmonary disease (COPD), and other respiratory disorders. Since inflammatory biomarkers such as IL-6, C-reactive protein (CRP), and TNF-α play a role in systemic inflammation, dietary patterns that reduce these markers may support overall lung health. A systematic review of RCTs analyzed data from 22 studies to determine the effects of diets such as the Mediterranean, DASH (Dietary Approaches to Stop Hypertension), and vegetarian/vegan diets. Inflammatory biomarkers, including CRP, IL-6, IL-1β, and TNF-α, were used as primary outcome measures. 95 The study found that the Mediterranean diet demonstrated the most significant reductions in these inflammatory biomarkers.
Other dietary patterns, such as DASH and vegetarian/vegan diets, were associated with moderate reductions in CRP but lacked significant effects on other biomarkers. These findings suggest the Mediterranean diet’s unique combination of plant-based foods, healthy fats, and moderate animal product intake is particularly effective in mitigating chronic inflammation. The anti-inflammatory effects are attributed to its high antioxidant potential, derived from components like olive oil, fruits, vegetables, and whole grains. These foods may suppress proinflammatory mediators, enhance antioxidant defenses, and modulate immune pathways.
Gut Health and Immunity
The Gut Immune Connection
The mammalian intestine hosts over a trillion microorganisms collectively known as the “gut microbiota,” a microbial community that has co-evolved with its host to establish a mutually beneficial relationship. Growing evidence suggests that the gut microbiota is essential for the development of the immune system and plays a pivotal role in defending the host against pathogens and in respiratory health.96 The gut microbiota influences the immune response by interacting with both the innate and adaptive immune systems. Recent research has also highlighted the gut-lung axis, a bidirectional communication pathway between the gut and the lungs, which influences respiratory health and disease.
Gut microbiota-derived metabolites, such as short-chain fatty acids (SCFAs), play a crucial role in modulating immune responses in the lungs. These metabolites can suppress lung inflammation and influence the severity of respiratory diseases by activating specific receptors.97
The innate immune response is influenced through various mechanisms, including the production of antimicrobial peptides, activation of inflammasomes, and induction of cytokines like IL-22, IL-17, and IL-10. 98
Antimicrobial peptides, such as α-defensins and β-defensins, are key players in this defense system. Produced by Paneth cells in response to bacterial signals, α-defensins disrupt microbial membranes, while β-defensins, regulated by microbiota via pathways like NF-κB, exhibit strong antimicrobial activity. C-type lectins further enhance protection by preventing bacterial colonization. These peptides collectively form a robust line of defense against intestinal pathogens.
Inflammasomes, multi-protein complexes activated by gut microbiota, are another critical component of innate immunity. They respond to microbial threats by secreting proinflammatory cytokines such as IL-1β and IL-18. Metabolites derived from gut microbiota, including short-chain fatty acids (SCFAs) and ATP, stimulate inflammasome pathways.
The gut microbiota also regulates cytokine production, which is vital for immune function. IL-22 strengthens mucosal barrier integrity and promotes antimicrobial protein expression, often stimulated by microbiota-derived metabolites produced by Lactobacillus species. Similarly, IL-17 aids in neutrophil recruitment and AMP production, with its expression influenced by bacteria like Bacteroides. IL-10, an anti-inflammatory cytokine, plays a central role in maintaining immune balance, produced by strains such as Clostridium butyricum.
The gut microbiota is also shown to significantly influence adaptive immunity by modulating T cell responses and maintaining immune balance. 99
The gut microbiota plays a critical role in the differentiation and function of T cells, particularly in the balance between pro-inflammatory and regulatory responses. Commensal bacteria such as Bacteroides fragilis promote regulatory T cell (Treg) development, which is essential for immune tolerance and preventing overactive immune responses.
The gut microbiota indirectly affects adaptive immunity through its interaction with antigen-presenting cells (APCs), such as dendritic cells. By influencing APC function, the microbiota helps determine whether T cells mount an inflammatory response or adopt a regulatory phenotype. This is essential for maintaining immune tolerance to non-pathogenic antigens in the gut.100
Dysbiosis, a disruption or imbalance in the gut microbiota’s composition, diversity, or functionality, negatively impacts the host’s health and immune system. It has been linked to several respiratory diseases, including asthma, chronic obstructive pulmonary disease (COPD), and lung cancer.101 Dysbiosis is triggered by factors such as a poor diet (e.g., high-fat or low-fiber Western diets), stress, infection, and antibiotic use. It is characterized by a reduction in beneficial microbes, an overgrowth of pathogenic or pro-inflammatory strains, and diminished production of critical microbial metabolites like short-chain fatty acids (SCFAs) and tryptophan derivatives.102
This imbalance compromises gut barrier integrity, allowing microbial antigens and toxins to enter circulation and trigger systemic inflammation. Dysbiosis alters immune system dynamics, shifting the balance from regulatory T cells (Tregs) to pro-inflammatory Th17 cells, exacerbating conditions such as inflammatory bowel disease, rheumatoid arthritis, type 1 diabetes, and other autoimmune disorders. It also reduces colonization resistance, increasing susceptibility to infections like Clostridium difficile. Dysbiosis also affects systemic immunity through mechanisms like cytokine dysregulation, impaired antimicrobial peptide production, and altered contributing to diseases such as respiratory infections, and metabolic syndromes. 103
A systematic review examined 11 studies that reported gut microbiome modifications in patients with a proven or suspected respiratory tract infections, compared to healthy, matched controls. Collectively, reported gut microbiome modifications in patients with a respiratory tract infection compared to healthy controls were consistently reported as decreased in diversity with a depletion of Firmicutes, Lachnospiraceae, Ruminococcaceae and Ruminococcus and enrichment of Enterococcus. 104 Probiotics, and prebiotics are being explored as potential therapeutic interventions to restore gut microbiota balance and improve respiratory health. These interventions aim to enhance beneficial bacteria and modulate immune responses. 105Novel approaches, such as fecal microbiota transplantation and the use of microbial-derived metabolites, are also being investigated for their potential to improve lung health and manage respiratory disease. 106
Maintaining a balanced gut microbiota involves a combination of dietary choices, lifestyle habits, and careful management of factors that can disrupt gut health. A diverse diet rich in fiber, regular exercise and the use of probiotics are key strategies. Additionally, minimizing antibiotic use and managing stress are important for sustaining a healthy gut environment. 107
It was shown that the Mediterranean Diet enhances gut microbial diversity, boosts SCFA production, and reduces inflammation while a Western Diet, high in fats, refined sugars, and food additives, increases inflammatory species, and disrupts intestinal barrier function. High-fat and low-carb diets known as the Ketogenic Diet, can reduce beneficial microbes like Bifidobacterium and alter mucus layer integrity, potentially increasing inflammation. A Gluten-Free Diet, though essential for celiac disease, may reduce beneficial bacteria like Bifidobacterium and Lactobacillus while increasing pathogenic species. 108
Probiotics and Prebiotics
The International Scientific Association for Probiotics and Prebiotics defines probiotics as “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host”. Prebiotics on the other hand, are typically complex non-digestible carbohydrates (such as inulin, other fructo-oligosaccharides and galacto-oligosaccharides) that microorganisms in the gastrointestinal tract use as metabolic fuel.
The seven core genera of microbial organisms most often used in probiotic products are Lactobacillus, Bifidobacterium, Saccharomyces, Streptococcus, Enterococcus, Escherichia, and Bacillus. Several in vitro, in vivo, and clinical studies have confirmed that probiotics and prebiotics play a significant role in maintaining intestinal microorganism equilibrium and regulating immune response.
Modulation of Gut Microbiome: Probiotics modulate gut microbiota by stimulating beneficial bacteria, and inhibiting harmful bacteria. They promote a balanced microbiota by engaging in competitive exclusion, outcompeting harmful microbes for adhesion sites and essential nutrients. Additionally, some probiotic bacteria regulate stomach acidity, creating an environment unfavorable for pathogenic organisms. Additionally, they enhance gut barrier integrity and inhibit pathogen colonization, contributing to a healthy intestinal environment. 109 Prebiotics, therefore, provide benefits to the host, including strengthening the integrity of the intestinal mucosal barrier, increasing host mucosal immunity, lowering pH and the production of Short Chain Fatty Acids (SCFAs) and inhibiting the growth of pathogenic microorganisms. 110
Modulation of Immune Function: Probiotics enhance the immune functions by interacting with toll-like receptors (TLRs) to activate signaling pathways and increase anti-inflammatory cytokines. 111 Prebiotics were found to alleviate metabolic disorders, and regulate immune responses through cytokine modulation. 112
Sources of Probiotics:
Fermented foods are produced through the growth and metabolic activity of various live microbial cultures. Many of these foods are excellent sources of live and potentially beneficial microbes. However, some fermented foods, like sourdough bread and most commercial pickles, undergo processing after fermentation, leaving them without live cultures in their final form. On the other hand, commercial yogurts often contain probiotic microorganisms such as Lactobacillus bulgaricus and Streptococcus thermophilus.
The live microorganisms in many fermented foods, including yogurt, generally remain viable throughout their shelf life. However, they often do not survive the acidic environment of the stomach or the enzymatic and bile activity in the small intestine, which can prevent them from reaching the distal gut. In contrast, probiotic strains specifically included in yogurt and other foods are designed to survive intestinal transit.
Fermented foods like many cheeses, kimchi (fermented cabbage), kombucha (fermented tea), sauerkraut, miso (fermented soybean paste), pickles, and raw unfiltered apple cider vinegar contain live cultures but typically lack proven probiotic strains.
Some unfermented foods, including milk, juices, smoothies, cereals, nutrition bars, and infant formulas, are fortified with added microorganisms. Their probiotic status depends on the microorganism levels at consumption, their ability to survive intestinal transit, and whether the specific strains provide health benefits.
Probiotics are also available as dietary supplements in various forms, such as capsules, powders, and liquids. These products often contain mixed cultures of live microorganisms rather than single strains. Many commercial probiotics have not been thoroughly studied, making it challenging for consumers unfamiliar with probiotic research to identify evidence-based products. However, some organizations offer guidance on specific probiotics, including appropriate strains, doses, and formulations for preventing or managing specific health conditions.
Who are the Experts
Here are some prominent researchers and medical professionals specializing in key nutrients and food for immune function, along with their areas of expertise:
1. Dr. Harri Hemilä
Affiliation: University of Helsinki, Finland
Expertise: Vitamin C, particularly in the context of its effects on immunity and respiratory infections such as the common cold.
Key Work: Analyzing and synthesizing evidence about vitamin C’s health benefits
Contact: harri.hemila@helsinki.fi
Selected Publications:
Hemilä, Harri, and Elizabeth Chalker. 2023. “Vitamin C Reduces the Severity of Common Colds: A Meta-Analysis.” BMC Public Health 23 (1): 13. https://doi: 10.1186/s12889-023-17229-8.
Hemilä, Harri, and Elizabeth Chalker. 2022. “Bias Against Vitamin C in Mainstream Medicine: Examples from Trials of Vitamin C for Infections.” Life (Basel) 12 (1): 35. https://doi.org/10.3390/life12010062.
Hemilä, Harri, and Elizabeth Chalker. 2021. “Commentary: Vitamin C Supplementation for Prevention and Treatment of Pneumonia.” Frontiers in Medicine 7: 2. https://doi.org/10.3389/fmed.2020.595988.
Hemilä, Harri, and Annelies M. E. de Man. 2021. “Vitamin C and COVID-19.” Frontiers in Medicine 7: 3. https://doi.org/10.3389/fmed.2020.559811.
2. Dr. Adrian Martineau
Affiliation: The London school of Medicine and Dentistry, Queen Mary University of London
Expertise: Vitamin D, with a focus on understanding its role in human health and disease prevention
Key Work: Studying how vitamin D modulates the immune system, particularly in preventing and treating infections like tuberculosis, COVID-19, and acute respiratory infections including the potential of vitamin D supplementation in reducing susceptibility to tuberculosis.
Contact: a.martineau@qmul.ac.uk
Selected Publications:
Ganmaa, Davaasambuu, and Adrian R. Martineau. 2020. “Vitamin D Supplements for Prevention of Tuberculosis Infection and Disease.” New England Journal of Medicine 383 (4): 359–68. https://doi.org/10.1056/NEJMoa1915176.
Martineau, Adrian R., and Margherita T. Cantorna. 2022. “Vitamin D for COVID-19: Where Are We Now?” Nature Reviews Immunology 22: 529–30. https://doi.org/10.1038/s41577-022-00726-4.
Jolliffe, David A., and Adrian R. Martineau. 2022. “Vitamin D Supplements for Prevention of COVID-19 or Other Acute Respiratory Infections: A Phase 3 Randomized Controlled Trial (CORONAVIT).” BMJ 378: e071230. https://doi.org/10.1136/bmj-2022-071230.
Jolliffe, David A., and Adrian R. Martineau. 2021. “Vitamin D Supplementation to Prevent Acute Respiratory Infections: A Systematic Review and Meta-Analysis of Aggregate Data from Randomised Controlled Trials.” The Lancet Diabetes & Endocrinology 9 (5): 276–92. https://doi.org/10.1016/S2213-8587(21)00051-6.
Middelkoop, Keren, and Adrian R. Martineau. 2023. “Vitamin D Supplementation to Prevent Tuberculosis Infection in South African Schoolchildren: Multicentre Phase 3 Double-Blind Randomised Placebo-Controlled Trial (ViDiKids).” International Journal of Infectious Diseases S1201-9712(23): 562–63. https://doi.org/10.1016/j.ijid.2023.06.014.
Positions and Views of Worldwide Governmental Medical and Health Organizations
Nonprofits and Private Organizations
Social Media Contributors
Bibliography
Anand, Swadha, and Sharmila S. Mande. “Diet, Microbiota and Gut-Lung Connection.” Frontiers in Microbiology 9 (September 19, 2018): 2147. https://doi.org/10.3389/fmicb.2018.02147.
Arredondo, Miguel, Ronny Martínez, Marco T Núñez, Manuel Ruz, and Manuel Olivares. “Inhibition of Iron and Copper Uptake by Iron, Copper and Zinc.” Biological Research 39, no. 1 (2006). https://doi.org/10.4067/S0716-97602006000100011.
Ballini, Andrea, Ioannis Alexandros Charitos, Stefania Cantore, Skender Topi, Lucrezia Bottalico, and Luigi Santacroce. “About Functional Foods: The Probiotics and Prebiotics State of Art.” Antibiotics 12, no. 4 (March 23, 2023): 635. https://doi.org/10.3390/antibiotics12040635.
Barak, V., T. Halperin, and I. Kalickman. “The Effect of Sambucol, a Black Elderberry-Based, Natural Product, on the Production of Human Cytokines: I. Inflammatory Cytokines.” European Cytokine Network 12, no. 2 (2001): 290–96.
Bozonet, Stephanie M., and Anitra C. Carr. “The Role of Physiological Vitamin C Concentrations on Key Functions of Neutrophils Isolated from Healthy Individuals.” Nutrients 11, no. 6 (June 17, 2019): 1363. https://doi.org/10.3390/nu11061363.
Brown, Nick, Antti Juhani Kukka, and Andreas Mårtensson. “Efficacy of Zinc as Adjunctive Pneumonia Treatment in Children Aged 2 to 60 Months in Low-Income and Middle-Income Countries: A Systematic Review and Meta-Analysis.” BMJ Paediatrics Open 4, no. 1 (July 2020): e000662. https://doi.org/10.1136/bmjpo-2020-000662.
Calder, Philip, Anitra Carr, Adrian Gombart, and Manfred Eggersdorfer. “Optimal Nutritional Status for a Well-Functioning Immune System Is an Important Factor to Protect against Viral Infections.” Nutrients 12, no. 4 (April 23, 2020): 1181. https://doi.org/10.3390/nu12041181.
Cannell, J. J., R. Vieth, J. C. Umhau, M. F. Holick, W. B. Grant, S. Madronich, C. F. Garland, and E. Giovannucci. “Epidemic Influenza and Vitamin D.” Epidemiology and Infection 134, no. 6 (December 2006): 1129–40. https://doi.org/10.1017/S0950268806007175.
Carboo, Janet Adede, Robin Claire Dolman-Macleod, Linda Malan, and Martani Johanni Lombard. “High-Dose Oral Vitamin D Supplementation for Prevention of Infections in Children Aged 0 to 59 Months: A Systematic Review and Meta-Analysis.” Nutrition Reviews 82, no. 5 (April 12, 2024): 579–99. https://doi.org/10.1093/nutrit/nuad082.
Carr, Anitra, and Silvia Maggini. “Vitamin C and Immune Function.” Nutrients 9, no. 11 (November 3, 2017): 1211. https://doi.org/10.3390/nu9111211.
Carr, Anitra, and Margreet Vissers. “Synthetic or Food-Derived Vitamin C—Are They Equally Bioavailable?” Nutrients 5, no. 11 (October 28, 2013): 4284–4304. https://doi.org/10.3390/nu5114284.
Casas, Rosa, Emilio Sacanella, and Ramon Estruch. “The Immune Protective Effect of the Mediterranean Diet against Chronic Low-Grade Inflammatory Diseases.” Endocrine, Metabolic & Immune Disorders-Drug Targets 14, no. 4 (November 12, 2014): 245–54. https://doi.org/10.2174/1871530314666140922153350.
Castro-Quezada, Itandehui, Blanca Román-Viñas, and Lluís Serra-Majem. “The Mediterranean Diet and Nutritional Adequacy: A Review.” Nutrients 6, no. 1 (January 3, 2014): 231–48. https://doi.org/10.3390/nu6010231.
Chakhtoura, M., N. Napoli, and G. El Hajj Fuleihan. “Commentary: Myths and Facts on Vitamin D amidst the COVID-19 Pandemic.” Metabolism 109 (August 2020): 154276. https://doi.org/10.1016/j.metabol.2020.154276.
Cheng, Hong-Yu, Meng-Xia Ning, De-Kun Chen, and Wen-Tao Ma. “Interactions Between the Gut Microbiota and the Host Innate Immune Response Against Pathogens.” Frontiers in Immunology 10 (March 29, 2019): 607. https://doi.org/10.3389/fimmu.2019.00607.
Colotta, Francesco, Birger Jansson, and Fabrizio Bonelli. “Modulation of Inflammatory and Immune Responses by Vitamin D.” Journal of Autoimmunity 85 (December 2017): 78–97. https://doi.org/10.1016/j.jaut.2017.07.007.
Craddock, Joel C, Elizabeth P Neale, Gregory E Peoples, and Yasmine C Probst. “Vegetarian-Based Dietary Patterns and Their Relation with Inflammatory and Immune Biomarkers: A Systematic Review and Meta-Analysis.” Advances in Nutrition 10, no. 3 (May 2019): 433–51. https://doi.org/10.1093/advances/nmy103.
Dardenne, M. “Zinc and Immune Function.” European Journal of Clinical Nutrition 56, no. S3 (August 1, 2002): S20–23. https://doi.org/10.1038/sj.ejcn.1601479.
Doseděl, Martin, Eduard Jirkovský, Kateřina Macáková, Lenka Krčmová, Lenka Javorská, Jana Pourová, Laura Mercolini, et al. “Vitamin C—Sources, Physiological Role, Kinetics, Deficiency, Use, Toxicity, and Determination.” Nutrients 13, no. 2 (February 13, 2021): 615. https://doi.org/10.3390/nu13020615.
Estruch, Ramon. “Anti-Inflammatory Effects of the Mediterranean Diet: The Experience of the PREDIMED Study.” Proceedings of the Nutrition Society 69, no. 3 (August 2010): 333–40. https://doi.org/10.1017/S0029665110001539.
Galior, Kornelia, Stefan Grebe, and Ravinder Singh. “Development of Vitamin D Toxicity from Overcorrection of Vitamin D Deficiency: A Review of Case Reports.” Nutrients 10, no. 8 (July 24, 2018): 953. https://doi.org/10.3390/nu10080953.
García-Ordás, María Teresa, Natalia Arias, Carmen Benavides, Oscar García-Olalla, and José Alberto Benítez-Andrades. “Evaluation of Country Dietary Habits Using Machine Learning Techniques in Relation to Deaths from COVID-19.” Healthcare 8, no. 4 (September 29, 2020): 371. https://doi.org/10.3390/healthcare8040371.
Ginde, Adit A., Jonathan M. Mansbach, and Carlos A. Camargo. “Vitamin D, Respiratory Infections, and Asthma.” Current Allergy and Asthma Reports 9, no. 1 (January 2009): 81–87. https://doi.org/10.1007/s11882-009-0012-7.
Grijalva, Carlos G. “Antibiotic Prescription Rates for Acute Respiratory Tract Infections in US Ambulatory Settings.” JAMA 302, no. 7 (August 19, 2009): 758. https://doi.org/10.1001/jama.2009.1163.
Hall, Sannette C., and Devendra K. Agrawal. “Vitamin D and Bronchial Asthma: An Overview of Data From the Past 5 Years.” Clinical Therapeutics 39, no. 5 (May 2017): 917–29. https://doi.org/10.1016/j.clinthera.2017.04.002.
Hambidge, K. Michael, Leland V. Miller, Jamie E. Westcott, and Nancy F. Krebs. “Dietary Reference Intakes for Zinc May Require Adjustment for Phytate Intake Based upon Model Predictions.” The Journal of Nutrition 138, no. 12 (December 2008): 2363–66. https://doi.org/10.3945/jn.108.093823.
Haraguchi, Seiki, Mitsumi Ikeda, Satoshi Akagi, and Yuji Hirao. “Dynamic Changes in pStat3 Are Involved in Meiotic Spindle Assembly in Mouse Oocytes.” International Journal of Molecular Sciences 21, no. 4 (February 12, 2020): 1220. https://doi.org/10.3390/ijms21041220.
Harri, Harri. “Zinc Lozenges May Shorten the Duration of Colds: A Systematic Review.” The Open Respiratory Medicine Journal 5, no. 1 (June 23, 2011): 51–58. https://doi.org/10.2174/1874306401105010051.
Hawkins, Jessie, Colby Baker, Lindsey Cherry, and Elizabeth Dunne. “Black Elderberry (Sambucus Nigra) Supplementation Effectively Treats Upper Respiratory Symptoms: A Meta-Analysis of Randomized, Controlled Clinical Trials.” Complementary Therapies in Medicine 42 (February 2019): 361–65. https://doi.org/10.1016/j.ctim.2018.12.004.
Hemilä, Harri. “Vitamin C and Infections.” Nutrients 9, no. 4 (March 29, 2017): 339. https://doi.org/10.3390/nu9040339.
———. “Vitamin C May Alleviate Exercise-Induced Bronchoconstriction: A Meta-Analysis.” BMJ Open 3, no. 6 (June 2013): e002416. https://doi.org/10.1136/bmjopen-2012-002416.
Hemilä, Harri, Anitra Carr, and Elizabeth Chalker. “Vitamin C May Increase the Recovery Rate of Outpatient Cases of SARS-CoV-2 Infection by 70%: Reanalysis of the COVID A to Z Randomized Clinical Trial.” Frontiers in Immunology 12 (May 10, 2021): 674681. https://doi.org/10.3389/fimmu.2021.674681.
Hemilä, Harri, and Elizabeth Chalker. “Vitamin C for Preventing and Treating the Common Cold.” Edited by Cochrane Acute Respiratory Infections Group. Cochrane Database of Systematic Reviews 2013, no. 5 (January 31, 2013). https://doi.org/10.1002/14651858.CD000980.pub4.
———. “Vitamin C Reduces the Severity of Common Colds: A Meta-Analysis.” BMC Public Health 23, no. 1 (December 11, 2023): 2468. https://doi.org/10.1186/s12889-023-17229-8.
Hever, Julieanna. “Plant-Based Diets: A Physician’s Guide.” The Permanente Journal 20, no. 3 (September 2016): 15–082. https://doi.org/10.7812/TPP/15-082.
Hojyo, Shintaro, and Toshiyuki Fukada. “Roles of Zinc Signaling in the Immune System.” Journal of Immunology Research 2016 (2016): 1–21. https://doi.org/10.1155/2016/6762343.
Hunter, Jennifer, Susan Arentz, Joshua Goldenberg, Guoyan Yang, Jennifer Beardsley, Stephen P Myers, Dominik Mertz, and Stephen Leeder. “Zinc for the Prevention or Treatment of Acute Viral Respiratory Tract Infections in Adults: A Rapid Systematic Review and Meta-Analysis of Randomised Controlled Trials.” BMJ Open 11, no. 11 (October 2021): e047474. https://doi.org/10.1136/bmjopen-2020-047474.
“Issue Information.” Phytotherapy Research 35, no. 3 (March 2021): 1125–26. https://doi.org/10.1002/ptr.6753.
Jiang, Linlin, Guoqing Zhang, Ye Li, Guirong Shi, and Minhui Li. “Potential Application of Plant-Based Functional Foods in the Development of Immune Boosters.” Frontiers in Pharmacology 12 (April 20, 2021): 637782. https://doi.org/10.3389/fphar.2021.637782.
Jin, Xuting, Jiajia Ren, Ruohan Li, Ya Gao, Haoying Zhang, Jiamei Li, Jingjing Zhang, Xiaochuang Wang, and Gang Wang. “Global Burden of Upper Respiratory Infections in 204 Countries and Territories, from 1990 to 2019.” eClinicalMedicine 37 (July 2021): 100986. https://doi.org/10.1016/j.eclinm.2021.100986.
Jolliffe, David A, Carlos A Camargo, John D Sluyter, Mary Aglipay, John F Aloia, Davaasambuu Ganmaa, Peter Bergman, et al. “Vitamin D Supplementation to Prevent Acute Respiratory Infections: A Systematic Review and Meta-Analysis of Aggregate Data from Randomised Controlled Trials.” The Lancet Diabetes & Endocrinology 9, no. 5 (May 2021): 276–92. https://doi.org/10.1016/S2213-8587(21)00051-6.
Kaźmierczak-Barańska, Julia, Karolina Boguszewska, Angelika Adamus-Grabicka, and Bolesław T. Karwowski. “Two Faces of Vitamin C—Antioxidative and Pro-Oxidative Agent.” Nutrients 12, no. 5 (May 21, 2020): 1501. https://doi.org/10.3390/nu12051501.
Kendrick, Karla N., Hyunju Kim, Casey M. Rebholz, Elizabeth Selvin, Lyn M. Steffen, and Stephen P. Juraschek. “Plant-Based Diets and Risk of Hospitalization with Respiratory Infection: Results from the Atherosclerosis Risk in Communities (ARIC) Study.” Nutrients 15, no. 19 (October 5, 2023): 4265. https://doi.org/10.3390/nu15194265.
King, Janet C, Kenneth H Brown, Rosalind S Gibson, Nancy F Krebs, Nicola M Lowe, Jonathan H Siekmann, and Daniel J Raiten. “Biomarkers of Nutrition for Development (BOND)—Zinc Review.” The Journal of Nutrition 146, no. 4 (April 2016): 858S-885S. https://doi.org/10.3945/jn.115.220079.
Kıran, Tugba Raika, Onder Otlu, and Aysun Bay Karabulut. “Oxidative Stress and Antioxidants in Health and Disease.” Journal of Laboratory Medicine 47, no. 1 (February 23, 2023): 1–11. https://doi.org/10.1515/labmed-2022-0108.
Koelman, Liselot, Caue Egea Rodrigues, and Krasimira Aleksandrova. “Effects of Dietary Patterns on Biomarkers of Inflammation and Immune Responses: A Systematic Review and Meta-Analysis of Randomized Controlled Trials.” Advances in Nutrition 13, no. 1 (January 2022): 101–15. https://doi.org/10.1093/advances/nmab086.
Krawitz, Christian, Mobarak Abu Mraheil, Michael Stein, Can Imirzalioglu, Eugen Domann, Stephan Pleschka, and Torsten Hain. “Inhibitory Activity of a Standardized Elderberry Liquid Extract against Clinically-Relevant Human Respiratory Bacterial Pathogens and Influenza A and B Viruses.” BMC Complementary and Alternative Medicine 11, no. 1 (December 2011): 16. https://doi.org/10.1186/1472-6882-11-16.
Lassi, Zohra S, Anoosh Moin, and Zulfiqar A Bhutta. “Zinc Supplementation for the Prevention of Pneumonia in Children Aged 2 Months to 59 Months.” Edited by Cochrane Acute Respiratory Infections Group. Cochrane Database of Systematic Reviews 2017, no. 1 (December 4, 2016). https://doi.org/10.1002/14651858.CD005978.pub3.
Liu, Yue, Jiaqi Wang, and Changxin Wu. “Modulation of Gut Microbiota and Immune System by Probiotics, Pre-Biotics, and Post-Biotics.” Frontiers in Nutrition 8 (January 3, 2022): 634897. https://doi.org/10.3389/fnut.2021.634897.
Malihi, Z., Zhenqiang Wu, Carlene M.M. Lawes, and Robert Scragg. “Adverse Events from Large Dose Vitamin D Supplementation Taken for One Year or Longer.” The Journal of Steroid Biochemistry and Molecular Biology 188 (April 2019): 29–37. https://doi.org/10.1016/j.jsbmb.2018.12.002.
Malihi, Zarintaj, Zhenqiang Wu, Alistair W Stewart, Carlene Mm Lawes, and Robert Scragg. “Hypercalcemia, Hypercalciuria, and Kidney Stones in Long-Term Studies of Vitamin D Supplementation: A Systematic Review and Meta-Analysis.” The American Journal of Clinical Nutrition 104, no. 4 (October 2016): 1039–51. https://doi.org/10.3945/ajcn.116.134981.
Manning, Jared, Birgitta Mitchell, Daniel A. Appadurai, Arvind Shakya, Laura Jean Pierce, Hongfang Wang, Vincent Nganga, et al. “Vitamin C Promotes Maturation of T-Cells.” Antioxidants & Redox Signaling 19, no. 17 (December 10, 2013): 2054–67. https://doi.org/10.1089/ars.2012.4988.
Martineau, Adrian R, David A Jolliffe, Richard L Hooper, Lauren Greenberg, John F Aloia, Peter Bergman, Gal Dubnov-Raz, et al. “Vitamin D Supplementation to Prevent Acute Respiratory Tract Infections: Systematic Review and Meta-Analysis of Individual Participant Data.” BMJ, February 15, 2017, i6583. https://doi.org/10.1136/bmj.i6583.
Martins, Rui, Ana Rita Carlos, Faouzi Braza, Jessica A. Thompson, Patricia Bastos-Amador, Susana Ramos, and Miguel P. Soares. “Disease Tolerance as an Inherent Component of Immunity.” Annual Review of Immunology 37, no. 1 (April 26, 2019): 405–37. https://doi.org/10.1146/annurev-immunol-042718-041739.
Murphy, K, and C Weaver. “An Introduction to Immunobiology and Innate Immunity.” In Janeway’s Immunobiology, 9th ed., 1–35. Taylor & Francis, 2017.
Nations, S. P., P. J. Boyer, L. A. Love, M. F. Burritt, J. A. Butz, G. I. Wolfe, L. S. Hynan, J. Reisch, and J. R. Trivedi. “Denture Cream: An Unusual Source of Excess Zinc, Leading to Hypocupremia and Neurologic Disease.” Neurology 71, no. 9 (August 26, 2008): 639–43. https://doi.org/10.1212/01.wnl.0000312375.79881.94.
Nault, Daryl, Taryn A Machingo, Andrea G Shipper, Daniel A Antiporta, Candyce Hamel, Sahar Nourouzpour, Menelaos Konstantinidis, Erica Phillips, Elizabeth A Lipski, and L Susan Wieland. “Zinc for Prevention and Treatment of the Common Cold.” Edited by Cochrane Acute Respiratory Infections Group. Cochrane Database of Systematic Reviews 2024, no. 5 (May 9, 2024). https://doi.org/10.1002/14651858.CD014914.pub2.
Neufingerl, Nicole, and Ans Eilander. “Nutrient Intake and Status in Adults Consuming Plant-Based Diets Compared to Meat-Eaters: A Systematic Review.” Nutrients 14, no. 1 (December 23, 2021): 29. https://doi.org/10.3390/nu14010029.
Olczak-Pruc, Monika, Damian Swieczkowski, Jerzy R. Ladny, Michal Pruc, Raul Juarez-Vela, Zubaid Rafique, Frank W. Peacock, and Lukasz Szarpak. “Vitamin C Supplementation for the Treatment of COVID-19: A Systematic Review and Meta-Analysis.” Nutrients 14, no. 19 (October 10, 2022): 4217. https://doi.org/10.3390/nu14194217.
Padayatty, Sebastian J., Andrew Y. Sun, Qi Chen, Michael Graham Espey, Jeanne Drisko, and Mark Levine. “Vitamin C: Intravenous Use by Complementary and Alternative Medicine Practitioners and Adverse Effects.” Edited by Joel Joseph Gagnier. PLoS ONE 5, no. 7 (July 7, 2010): e11414. https://doi.org/10.1371/journal.pone.0011414.
Padhani, Zahra Ali, Zorays Moazzam, Alina Ashraf, Bilal Hasana, Rehana A Salam, Jai K Das, and Zulfiqar A Bhutta. “Vitamin C Supplementation for Prevention and Treatment of Pneumonia.” Edited by Cochrane Acute Respiratory Infections Group. Cochrane Database of Systematic Reviews 2021, no. 11 (November 18, 2021). https://doi.org/10.1002/14651858.CD013134.pub3.
Pehlivan, Fadime Eryılmaz. “Vitamin C: An Antioxidant Agent.” In Vitamin C, edited by Amal H. Hamza. InTech, 2017. https://doi.org/10.5772/intechopen.69660.
Rinninella, Emanuele, Marco Cintoni, Pauline Raoul, Loris Riccardo Lopetuso, Franco Scaldaferri, Gabriele Pulcini, Giacinto Abele Donato Miggiano, Antonio Gasbarrini, and Maria Cristina Mele. “Food Components and Dietary Habits: Keys for a Healthy Gut Microbiota Composition.” Nutrients 11, no. 10 (October 7, 2019): 2393. https://doi.org/10.3390/nu11102393.
Rinninella, Emanuele, Pauline Raoul, Marco Cintoni, Francesco Franceschi, Giacinto Abele Donato Miggiano, Antonio Gasbarrini, and Maria Cristina Mele. “What Is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases.” Microorganisms 7, no. 1 (January 10, 2019): 14. https://doi.org/10.3390/microorganisms7010014.
Senica, Mateja, Franci Stampar, Robert Veberic, and Maja Mikulic‐Petkovsek. “The Higher the Better? Differences in Phenolics and Cyanogenic Glycosides in Sambucus Nigra Leaves, Flowers and Berries from Different Altitudes.” Journal of the Science of Food and Agriculture 97, no. 8 (June 2017): 2623–32. https://doi.org/10.1002/jsfa.8085.
Sharma, Yogesh, Subodha Sumanadasa, Rashmi Shahi, Richard Woodman, Arduino A. Mangoni, Shailesh Bihari, and Campbell Thompson. “Efficacy and Safety of Vitamin C Supplementation in the Treatment of Community-Acquired Pneumonia: A Systematic Review and Meta-Analysis with Trial Sequential Analysis.” Scientific Reports 14, no. 1 (May 24, 2024): 11846. https://doi.org/10.1038/s41598-024-62571-5.
Sîrbu, Alexandru Constantin, Octavia Sabin, Ioana Corina Bocșan, Ștefan Cristian Vesa, and Anca Dana Buzoianu. “The Effect of Vitamin D Supplementation on the Length of Hospitalisation, Intensive Care Unit Admission, and Mortality in COVID-19—A Systematic Review and Meta-Analysis.” Nutrients 15, no. 15 (August 5, 2023): 3470. https://doi.org/10.3390/nu15153470.
Skalny, Anatoly, Lothar Rink, Olga Ajsuvakova, Michael Aschner, Viktor Gritsenko, Svetlana Alekseenko, Andrey Svistunov, et al. “Zinc and Respiratory Tract Infections: Perspectives for COVID‑19 (Review).” International Journal of Molecular Medicine, April 14, 2020. https://doi.org/10.3892/ijmm.2020.4575.
Sultan, M. Tauseef, Masood Sadiq Buttxs, Mir M. Nasir Qayyum, and Hafiz Ansar Rasul Suleria. “Immunity: Plants as Effective Mediators.” Critical Reviews in Food Science and Nutrition 54, no. 10 (January 2014): 1298–1308. https://doi.org/10.1080/10408398.2011.633249.
Tarasov, Andrei V., Rofail S. Rakhmanov, Elena S. Bogomolova, Ludmila A. Perminova, and Zhanna L. Malakhova. “The Role of Plant-Based Protein Functional Food in Preventing Acute Respiratory Disease: A Case Study.” Nutrients 13, no. 6 (June 20, 2021): 2116. https://doi.org/10.3390/nu13062116.
Tiralongo, Evelin, Shirley Wee, and Rodney Lea. “Elderberry Supplementation Reduces Cold Duration and Symptoms in Air-Travellers: A Randomized, Double-Blind Placebo-Controlled Clinical Trial.” Nutrients 8, no. 4 (March 24, 2016): 182. https://doi.org/10.3390/nu8040182.
Torabian, Golnoosh, Peter Valtchev, Qayyum Adil, and Fariba Dehghani. “Anti-Influenza Activity of Elderberry (Sambucus Nigra).” Journal of Functional Foods 54 (March 2019): 353–60. https://doi.org/10.1016/j.jff.2019.01.031.
Tourkochristou, Evanthia, Christos Triantos, and Athanasia Mouzaki. “The Influence of Nutritional Factors on Immunological Outcomes.” Frontiers in Immunology 12 (May 31, 2021): 665968. https://doi.org/10.3389/fimmu.2021.665968.
Trivedi, Riddhi, and Kalyani Barve. “Gut Microbiome a Promising Target for Management of Respiratory Diseases.” Biochemical Journal 477, no. 14 (July 31, 2020): 2679–96. https://doi.org/10.1042/BCJ20200426.
Verma, Anjali, Tannu Bhagchandani, Ankita Rai, Nikita, Urvinder Kaur Sardarni, Neel Sarovar Bhavesh, Sameer Gulati, Rupali Malik, and Ravi Tandon. “Short-Chain Fatty Acid (SCFA) as a Connecting Link between Microbiota and Gut-Lung Axis─A Potential Therapeutic Intervention to Improve Lung Health.” ACS Omega 9, no. 13 (April 2, 2024): 14648–71. https://doi.org/10.1021/acsomega.3c05846.
Wakeman, Michael. “A Literature Review of the Potential Impact of Medication on Vitamin D Status.” Risk Management and Healthcare Policy Volume 14 (August 2021): 3357–81. https://doi.org/10.2147/RMHP.S316897.
Wegmüller, Rita, Fabian Tay, Christophe Zeder, Marica Brnić, and Richard F. Hurrell. “Zinc Absorption by Young Adults from Supplemental Zinc Citrate Is Comparable with That from Zinc Gluconate and Higher than from Zinc Oxide.” The Journal of Nutrition 144, no. 2 (February 2014): 132–36. https://doi.org/10.3945/jn.113.181487.
Wen, Chengli, Yuan Li, Qinxue Hu, Hui Liu, Xinxin Xu, and Muhan Lü. “IV Vitamin C in Sepsis: A Latest Systematic Review and Meta-Analysis.” Edited by Ahmed Abdelwahab. International Journal of Clinical Practice 2023 (January 24, 2023): 1–15. https://doi.org/10.1155/2023/6733465.
Wessels, Inga, Henrike Josephine Fischer, and Lothar Rink. “Dietary and Physiological Effects of Zinc on the Immune System.” Annual Review of Nutrition 41, no. 1 (October 11, 2021): 133–75. https://doi.org/10.1146/annurev-nutr-122019-120635.
Wessels, Inga, Martina Maywald, and Lothar Rink. “Zinc as a Gatekeeper of Immune Function.” Nutrients 9, no. 12 (November 25, 2017): 1286. https://doi.org/10.3390/nu9121286.
Wharton, Robert C., Jing Gennie Wang, Yuni Choi, Elliot Eisenberg, Mariah K. Jackson, Corrine Hanson, Bian Liu, et al. “Associations of a Plant-Centered Diet and Lung Function across Early to Mid-Adulthood: The CARDIA Lung Study.” Respiratory Research 25, no. 1 (March 11, 2024): 122. https://doi.org/10.1186/s12931-023-02632-x.
Wieland, L. Susan, Vanessa Piechotta, Termeh Feinberg, Emilie Ludeman, Brian Hutton, Salmaan Kanji, Dugald Seely, and Chantelle Garritty. “Elderberry for Prevention and Treatment of Viral Respiratory Illnesses: A Systematic Review.” BMC Complementary Medicine and Therapies 21, no. 1 (December 2021): 112. https://doi.org/10.1186/s12906-021-03283-5.
Woodall, Claire A., Luke J. McGeoch, Alastair D. Hay, and Ashley Hammond. “Respiratory Tract Infections and Gut Microbiome Modifications: A Systematic Review.” Edited by Aran Singanayagam. PLOS ONE 17, no. 1 (January 13, 2022): e0262057. https://doi.org/10.1371/journal.pone.0262057.
Wu, Dayong, Erin D. Lewis, Munyong Pae, and Simin Nikbin Meydani. “Nutritional Modulation of Immune Function: Analysis of Evidence, Mechanisms, and Clinical Relevance.” Frontiers in Immunology 9 (January 15, 2019): 3160. https://doi.org/10.3389/fimmu.2018.03160.
Wu, Hsin-Jung, and Eric Wu. “The Role of Gut Microbiota in Immune Homeostasis and Autoimmunity.” Gut Microbes 3, no. 1 (January 2012): 4–14. https://doi.org/10.4161/gmic.19320.
Xing, Xin, Min Xu, Lijun Yang, Wenqian Zhang, Xiaolin Niu, and Dengfeng Gao. “The Efficacy of Intravenous Vitamin C in Critically Ill Patients: A Meta-Analysis of Randomized Controlled Trials.” Clinical Nutrition 40, no. 5 (May 2021): 2630–39. https://doi.org/10.1016/j.clnu.2021.03.007.
Yoo, Ji, Maureen Groer, Samia Dutra, Anujit Sarkar, and Daniel McSkimming. “Gut Microbiota and Immune System Interactions.” Microorganisms 8, no. 10 (October 15, 2020): 1587. https://doi.org/10.3390/microorganisms8101587.
Zakay-Rones, Z, E Thom, T Wollan, and J Wadstein. “Randomized Study of the Efficacy and Safety of Oral Elderberry Extract in the Treatment of Influenza A and B Virus Infections.” Journal of International Medical Research 32, no. 2 (April 2004): 132–40. https://doi.org/10.1177/147323000403200205.
Zhou, Pengjun, Chunlan Chen, Sandip Patil, and Shaowei Dong. “Unveiling the Therapeutic Symphony of Probiotics, Prebiotics, and Postbiotics in Gut-Immune Harmony.” Frontiers in Nutrition 11 (February 8, 2024): 1355542. https://doi.org/10.3389/fnut.2024.1355542.
Zisi, Dimitra, Anna Challa, and Alexandros Makis. “The Association between Vitamin D Status and Infectious Diseases of the Respiratory System in Infancy and Childhood.” Hormones 18, no. 4 (December 2019): 353–63. https://doi.org/10.1007/s42000-019-00155-z.
Evanthia Tourkochristou, Christos Triantos, and Athanasia Mouzaki, “The Influence of Nutritional Factors on Immunological Outcomes,” Frontiers in Immunology 12 (May 31, 2021): 665968, https://doi.org/10.3389/fimmu.2021.665968.↩︎
Dayong Wu et al., “Nutritional Modulation of Immune Function: Analysis of Evidence, Mechanisms, and Clinical Relevance,” Frontiers in Immunology 9 (January 15, 2019): 3160, https://doi.org/10.3389/fimmu.2018.03160.↩︎
Rui Martins et al., “Disease Tolerance as an Inherent Component of Immunity,” Annual Review of Immunology 37, no. 1 (April 26, 2019): 405–37, https://doi.org/10.1146/annurev-immunol-042718-041739.↩︎
Xuting Jin et al., “Global Burden of Upper Respiratory Infections in 204 Countries and Territories, from 1990 to 2019,” eClinicalMedicine 37 (July 2021): 100986, https://doi.org/10.1016/j.eclinm.2021.100986.↩︎
Carlos G. Grijalva, “Antibiotic Prescription Rates for Acute Respiratory Tract Infections in US Ambulatory Settings,” JAMA 302, no. 7 (August 19, 2009): 758, https://doi.org/10.1001/jama.2009.1163.↩︎
K Murphy and C Weaver, “An Introduction to Immunobiology and Innate Immunity,” in Janeway’s Immunobiology, 9th ed. (Taylor & Francis, 2017), 1–35.↩︎
Philip Calder et al., “Optimal Nutritional Status for a Well-Functioning Immune System Is an Important Factor to Protect against Viral Infections,” Nutrients 12, no. 4 (April 23, 2020): 1181, https://doi.org/10.3390/nu12041181.↩︎
Harri Hemilä, “Vitamin C and Infections,” Nutrients 9, no. 4 (March 29, 2017): 339, https://doi.org/10.3390/nu9040339.↩︎
Fadime Eryılmaz Pehlivan, “Vitamin C: An Antioxidant Agent,” in Vitamin C, ed. Amal H. Hamza (InTech, 2017), https://doi.org/10.5772/intechopen.69660.↩︎
Tugba Raika Kıran, Onder Otlu, and Aysun Bay Karabulut, “Oxidative Stress and Antioxidants in Health and Disease,” Journal of Laboratory Medicine 47, no. 1 (February 23, 2023): 1–11, https://doi.org/10.1515/labmed-2022-0108.↩︎
Kıran, Otlu, and Karabulut.↩︎
Julia Kaźmierczak-Barańska et al., “Two Faces of Vitamin C—Antioxidative and Pro-Oxidative Agent,” Nutrients 12, no. 5 (May 21, 2020): 1501, https://doi.org/10.3390/nu12051501.↩︎
Anitra Carr and Silvia Maggini, “Vitamin C and Immune Function,” Nutrients 9, no. 11 (November 3, 2017): 1211, https://doi.org/10.3390/nu9111211.↩︎
Stephanie M. Bozonet and Anitra C. Carr, “The Role of Physiological Vitamin C Concentrations on Key Functions of Neutrophils Isolated from Healthy Individuals,” Nutrients 11, no. 6 (June 17, 2019): 1363, https://doi.org/10.3390/nu11061363.↩︎
Jared Manning et al., “Vitamin C Promotes Maturation of T-Cells,” Antioxidants & Redox Signaling 19, no. 17 (December 10, 2013): 2054–67, https://doi.org/10.1089/ars.2012.4988.↩︎
Carr and Maggini, “Vitamin C and Immune Function.”↩︎
Harri Hemilä and Elizabeth Chalker, “Vitamin C for Preventing and Treating the Common Cold,” ed. Cochrane Acute Respiratory Infections Group, Cochrane Database of Systematic Reviews 2013, no. 5 (January 31, 2013), https://doi.org/10.1002/14651858.CD000980.pub4.↩︎
Harri Hemilä and Elizabeth Chalker, “Vitamin C Reduces the Severity of Common Colds: A Meta-Analysis,” BMC Public Health 23, no. 1 (December 11, 2023): 2468, https://doi.org/10.1186/s12889-023-17229-8.↩︎
Hemilä and Chalker, “Vitamin C for Preventing and Treating the Common Cold.”↩︎
Harri Hemilä, “Vitamin C May Alleviate Exercise-Induced Bronchoconstriction: A Meta-Analysis,” BMJ Open 3, no. 6 (June 2013): e002416, https://doi.org/10.1136/bmjopen-2012-002416.↩︎
Yogesh Sharma et al., “Efficacy and Safety of Vitamin C Supplementation in the Treatment of Community-Acquired Pneumonia: A Systematic Review and Meta-Analysis with Trial Sequential Analysis,” Scientific Reports 14, no. 1 (May 24, 2024): 11846, https://doi.org/10.1038/s41598-024-62571-5.↩︎
Monika Olczak-Pruc et al., “Vitamin C Supplementation for the Treatment of COVID-19: A Systematic Review and Meta-Analysis,” Nutrients 14, no. 19 (October 10, 2022): 4217, https://doi.org/10.3390/nu14194217.↩︎
Zahra Ali Padhani et al., “Vitamin C Supplementation for Prevention and Treatment of Pneumonia,” ed. Cochrane Acute Respiratory Infections Group, Cochrane Database of Systematic Reviews 2021, no. 11 (November 18, 2021), https://doi.org/10.1002/14651858.CD013134.pub3.↩︎
Chengli Wen et al., “IV Vitamin C in Sepsis: A Latest Systematic Review and Meta-Analysis,” ed. Ahmed Abdelwahab, International Journal of Clinical Practice 2023 (January 24, 2023): 1–15, https://doi.org/10.1155/2023/6733465.↩︎
Xin Xing et al., “The Efficacy of Intravenous Vitamin C in Critically Ill Patients: A Meta-Analysis of Randomized Controlled Trials,” Clinical Nutrition 40, no. 5 (May 2021): 2630–39, https://doi.org/10.1016/j.clnu.2021.03.007.↩︎
Xing et al.↩︎
Harri Hemilä, Anitra Carr, and Elizabeth Chalker, “Vitamin C May Increase the Recovery Rate of Outpatient Cases of SARS-CoV-2 Infection by 70%: Reanalysis of the COVID A to Z Randomized Clinical Trial,” Frontiers in Immunology 12 (May 10, 2021): 674681, https://doi.org/10.3389/fimmu.2021.674681.↩︎
Anitra Carr and Margreet Vissers, “Synthetic or Food-Derived Vitamin C—Are They Equally Bioavailable?,” Nutrients 5, no. 11 (October 28, 2013): 4284–4304, https://doi.org/10.3390/nu5114284.↩︎
Carr and Vissers.↩︎
Martin Doseděl et al., “Vitamin C—Sources, Physiological Role, Kinetics, Deficiency, Use, Toxicity, and Determination,” Nutrients 13, no. 2 (February 13, 2021): 615, https://doi.org/10.3390/nu13020615.↩︎
Sebastian J. Padayatty et al., “Vitamin C: Intravenous Use by Complementary and Alternative Medicine Practitioners and Adverse Effects,” ed. Joel Joseph Gagnier, PLoS ONE 5, no. 7 (July 7, 2010): e11414, https://doi.org/10.1371/journal.pone.0011414.↩︎
Francesco Colotta, Birger Jansson, and Fabrizio Bonelli, “Modulation of Inflammatory and Immune Responses by Vitamin D,” Journal of Autoimmunity 85 (December 2017): 78–97, https://doi.org/10.1016/j.jaut.2017.07.007.↩︎
Sannette C. Hall and Devendra K. Agrawal, “Vitamin D and Bronchial Asthma: An Overview of Data From the Past 5 Years,” Clinical Therapeutics 39, no. 5 (May 2017): 917–29, https://doi.org/10.1016/j.clinthera.2017.04.002.↩︎
J. J. Cannell et al., “Epidemic Influenza and Vitamin D,” Epidemiology and Infection 134, no. 6 (December 2006): 1129–40, https://doi.org/10.1017/S0950268806007175.↩︎
Adrian R Martineau et al., “Vitamin D Supplementation to Prevent Acute Respiratory Tract Infections: Systematic Review and Meta-Analysis of Individual Participant Data,” BMJ, February 15, 2017, i6583, https://doi.org/10.1136/bmj.i6583.↩︎
Martineau et al.↩︎
David A Jolliffe et al., “Vitamin D Supplementation to Prevent Acute Respiratory Infections: A Systematic Review and Meta-Analysis of Aggregate Data from Randomised Controlled Trials,” The Lancet Diabetes & Endocrinology 9, no. 5 (May 2021): 276–92, https://doi.org/10.1016/S2213-8587(21)00051-6.↩︎
Martineau et al., “Vitamin D Supplementation to Prevent Acute Respiratory Tract Infections.”↩︎
Jolliffe et al., “Vitamin D Supplementation to Prevent Acute Respiratory Infections.”↩︎
Alexandru Constantin Sîrbu et al., “The Effect of Vitamin D Supplementation on the Length of Hospitalisation, Intensive Care Unit Admission, and Mortality in COVID-19—A Systematic Review and Meta-Analysis,” Nutrients 15, no. 15 (August 5, 2023): 3470, https://doi.org/10.3390/nu15153470.↩︎
Janet Adede Carboo et al., “High-Dose Oral Vitamin D Supplementation for Prevention of Infections in Children Aged 0 to 59 Months: A Systematic Review and Meta-Analysis,” Nutrition Reviews 82, no. 5 (April 12, 2024): 579–99, https://doi.org/10.1093/nutrit/nuad082.↩︎
Dimitra Zisi, Anna Challa, and Alexandros Makis, “The Association between Vitamin D Status and Infectious Diseases of the Respiratory System in Infancy and Childhood,” Hormones 18, no. 4 (December 2019): 353–63, https://doi.org/10.1007/s42000-019-00155-z.↩︎
Adit A. Ginde, Jonathan M. Mansbach, and Carlos A. Camargo, “Vitamin D, Respiratory Infections, and Asthma,” Current Allergy and Asthma Reports 9, no. 1 (January 2009): 81–87, https://doi.org/10.1007/s11882-009-0012-7.↩︎
M. Chakhtoura, N. Napoli, and G. El Hajj Fuleihan, “Commentary: Myths and Facts on Vitamin D amidst the COVID-19 Pandemic,” Metabolism 109 (August 2020): 154276, https://doi.org/10.1016/j.metabol.2020.154276.↩︎
Z. Malihi et al., “Adverse Events from Large Dose Vitamin D Supplementation Taken for One Year or Longer,” The Journal of Steroid Biochemistry and Molecular Biology 188 (April 2019): 29–37, https://doi.org/10.1016/j.jsbmb.2018.12.002.↩︎
Zarintaj Malihi et al., “Hypercalcemia, Hypercalciuria, and Kidney Stones in Long-Term Studies of Vitamin D Supplementation: A Systematic Review and Meta-Analysis,” The American Journal of Clinical Nutrition 104, no. 4 (October 2016): 1039–51, https://doi.org/10.3945/ajcn.116.134981.↩︎
Kornelia Galior, Stefan Grebe, and Ravinder Singh, “Development of Vitamin D Toxicity from Overcorrection of Vitamin D Deficiency: A Review of Case Reports,” Nutrients 10, no. 8 (July 24, 2018): 953, https://doi.org/10.3390/nu10080953.↩︎
Michael Wakeman, “A Literature Review of the Potential Impact of Medication on Vitamin D Status,” Risk Management and Healthcare Policy Volume 14 (August 2021): 3357–81, https://doi.org/10.2147/RMHP.S316897.↩︎
Inga Wessels, Martina Maywald, and Lothar Rink, “Zinc as a Gatekeeper of Immune Function,” Nutrients 9, no. 12 (November 25, 2017): 1286, https://doi.org/10.3390/nu9121286.↩︎
Shintaro Hojyo and Toshiyuki Fukada, “Roles of Zinc Signaling in the Immune System,” Journal of Immunology Research 2016 (2016): 1–21, https://doi.org/10.1155/2016/6762343.↩︎
Inga Wessels, Henrike Josephine Fischer, and Lothar Rink, “Dietary and Physiological Effects of Zinc on the Immune System,” Annual Review of Nutrition 41, no. 1 (October 11, 2021): 133–75, https://doi.org/10.1146/annurev-nutr-122019-120635.↩︎
Wessels, Maywald, and Rink, “Zinc as a Gatekeeper of Immune Function.”↩︎
Wessels, Fischer, and Rink, “Dietary and Physiological Effects of Zinc on the Immune System.”↩︎
M Dardenne, “Zinc and Immune Function,” European Journal of Clinical Nutrition 56, no. S3 (August 1, 2002): S20–23, https://doi.org/10.1038/sj.ejcn.1601479.↩︎
Harri Harri, “Zinc Lozenges May Shorten the Duration of Colds: A Systematic Review,” The Open Respiratory Medicine Journal 5, no. 1 (June 23, 2011): 51–58, https://doi.org/10.2174/1874306401105010051.↩︎
Jennifer Hunter et al., “Zinc for the Prevention or Treatment of Acute Viral Respiratory Tract Infections in Adults: A Rapid Systematic Review and Meta-Analysis of Randomised Controlled Trials,” BMJ Open 11, no. 11 (October 2021): e047474, https://doi.org/10.1136/bmjopen-2020-047474.↩︎
Hunter et al.↩︎
Hunter et al.↩︎
Harri, “Zinc Lozenges May Shorten the Duration of Colds.”↩︎
Daryl Nault et al., “Zinc for Prevention and Treatment of the Common Cold,” ed. Cochrane Acute Respiratory Infections Group, Cochrane Database of Systematic Reviews 2024, no. 5 (May 9, 2024), https://doi.org/10.1002/14651858.CD014914.pub2.↩︎
Zohra S Lassi, Anoosh Moin, and Zulfiqar A Bhutta, “Zinc Supplementation for the Prevention of Pneumonia in Children Aged 2 Months to 59 Months,” ed. Cochrane Acute Respiratory Infections Group, Cochrane Database of Systematic Reviews 2017, no. 1 (December 4, 2016), https://doi.org/10.1002/14651858.CD005978.pub3.↩︎
Nick Brown, Antti Juhani Kukka, and Andreas Mårtensson, “Efficacy of Zinc as Adjunctive Pneumonia Treatment in Children Aged 2 to 60 Months in Low-Income and Middle-Income Countries: A Systematic Review and Meta-Analysis,” BMJ Paediatrics Open 4, no. 1 (July 2020): e000662, https://doi.org/10.1136/bmjpo-2020-000662.↩︎
Anatoly Skalny et al., “Zinc and Respiratory Tract Infections: Perspectives for COVID‑19 (Review),” International Journal of Molecular Medicine, April 14, 2020, https://doi.org/10.3892/ijmm.2020.4575.↩︎
Skalny et al.↩︎
K. Michael Hambidge et al., “Dietary Reference Intakes for Zinc May Require Adjustment for Phytate Intake Based upon Model Predictions,” The Journal of Nutrition 138, no. 12 (December 2008): 2363–66, https://doi.org/10.3945/jn.108.093823.↩︎
Janet C King et al., “Biomarkers of Nutrition for Development (BOND)—Zinc Review,” The Journal of Nutrition 146, no. 4 (April 2016): 858S-885S, https://doi.org/10.3945/jn.115.220079.↩︎
Rita Wegmüller et al., “Zinc Absorption by Young Adults from Supplemental Zinc Citrate Is Comparable with That from Zinc Gluconate and Higher than from Zinc Oxide,” The Journal of Nutrition 144, no. 2 (February 2014): 132–36, https://doi.org/10.3945/jn.113.181487.↩︎
Miguel Arredondo et al., “Inhibition of Iron and Copper Uptake by Iron, Copper and Zinc,” Biological Research 39, no. 1 (2006), https://doi.org/10.4067/S0716-97602006000100011.↩︎
King et al., “Biomarkers of Nutrition for Development (BOND)—Zinc Review.”↩︎
Wegmüller et al., “Zinc Absorption by Young Adults from Supplemental Zinc Citrate Is Comparable with That from Zinc Gluconate and Higher than from Zinc Oxide.”↩︎
S. P. Nations et al., “Denture Cream: An Unusual Source of Excess Zinc, Leading to Hypocupremia and Neurologic Disease,” Neurology 71, no. 9 (August 26, 2008): 639–43, https://doi.org/10.1212/01.wnl.0000312375.79881.94.↩︎
Z Zakay-Rones et al., “Randomized Study of the Efficacy and Safety of Oral Elderberry Extract in the Treatment of Influenza A and B Virus Infections,” Journal of International Medical Research 32, no. 2 (April 2004): 132–40, https://doi.org/10.1177/147323000403200205.↩︎
Christian Krawitz et al., “Inhibitory Activity of a Standardized Elderberry Liquid Extract against Clinically-Relevant Human Respiratory Bacterial Pathogens and Influenza A and B Viruses,” BMC Complementary and Alternative Medicine 11, no. 1 (December 2011): 16, https://doi.org/10.1186/1472-6882-11-16.↩︎
Golnoosh Torabian et al., “Anti-Influenza Activity of Elderberry (Sambucus Nigra),” Journal of Functional Foods 54 (March 2019): 353–60, https://doi.org/10.1016/j.jff.2019.01.031.↩︎
V. Barak, T. Halperin, and I. Kalickman, “The Effect of Sambucol, a Black Elderberry-Based, Natural Product, on the Production of Human Cytokines: I. Inflammatory Cytokines,” European Cytokine Network 12, no. 2 (2001): 290–96.↩︎
Barak, Halperin, and Kalickman.↩︎
Seiki Haraguchi et al., “Dynamic Changes in pStat3 Are Involved in Meiotic Spindle Assembly in Mouse Oocytes,” International Journal of Molecular Sciences 21, no. 4 (February 12, 2020): 1220, https://doi.org/10.3390/ijms21041220.↩︎
Evelin Tiralongo, Shirley Wee, and Rodney Lea, “Elderberry Supplementation Reduces Cold Duration and Symptoms in Air-Travellers: A Randomized, Double-Blind Placebo-Controlled Clinical Trial,” Nutrients 8, no. 4 (March 24, 2016): 182, https://doi.org/10.3390/nu8040182.↩︎
L. Susan Wieland et al., “Elderberry for Prevention and Treatment of Viral Respiratory Illnesses: A Systematic Review,” BMC Complementary Medicine and Therapies 21, no. 1 (December 2021): 112, https://doi.org/10.1186/s12906-021-03283-5.↩︎
Jessie Hawkins et al., “Black Elderberry (Sambucus Nigra) Supplementation Effectively Treats Upper Respiratory Symptoms: A Meta-Analysis of Randomized, Controlled Clinical Trials,” Complementary Therapies in Medicine 42 (February 2019): 361–65, https://doi.org/10.1016/j.ctim.2018.12.004.↩︎
“Issue Information,” Phytotherapy Research 35, no. 3 (March 2021): 1125–26, https://doi.org/10.1002/ptr.6753.↩︎
Mateja Senica et al., “The Higher the Better? Differences in Phenolics and Cyanogenic Glycosides in Sambucus Nigra Leaves, Flowers and Berries from Different Altitudes,” Journal of the Science of Food and Agriculture 97, no. 8 (June 2017): 2623–32, https://doi.org/10.1002/jsfa.8085.↩︎
Andrei V. Tarasov et al., “The Role of Plant-Based Protein Functional Food in Preventing Acute Respiratory Disease: A Case Study,” Nutrients 13, no. 6 (June 20, 2021): 2116, https://doi.org/10.3390/nu13062116.↩︎
Robert C. Wharton et al., “Associations of a Plant-Centered Diet and Lung Function across Early to Mid-Adulthood: The CARDIA Lung Study,” Respiratory Research 25, no. 1 (March 11, 2024): 122, https://doi.org/10.1186/s12931-023-02632-x.↩︎
Julieanna Hever, “Plant-Based Diets: A Physician’s Guide,” The Permanente Journal 20, no. 3 (September 2016): 15–082, https://doi.org/10.7812/TPP/15-082.↩︎
Linlin Jiang et al., “Potential Application of Plant-Based Functional Foods in the Development of Immune Boosters,” Frontiers in Pharmacology 12 (April 20, 2021): 637782, https://doi.org/10.3389/fphar.2021.637782.↩︎
Nicole Neufingerl and Ans Eilander, “Nutrient Intake and Status in Adults Consuming Plant-Based Diets Compared to Meat-Eaters: A Systematic Review,” Nutrients 14, no. 1 (December 23, 2021): 29, https://doi.org/10.3390/nu14010029.↩︎
Joel C Craddock et al., “Vegetarian-Based Dietary Patterns and Their Relation with Inflammatory and Immune Biomarkers: A Systematic Review and Meta-Analysis,” Advances in Nutrition 10, no. 3 (May 2019): 433–51, https://doi.org/10.1093/advances/nmy103.↩︎
M. Tauseef Sultan et al., “Immunity: Plants as Effective Mediators,” Critical Reviews in Food Science and Nutrition 54, no. 10 (January 2014): 1298–1308, https://doi.org/10.1080/10408398.2011.633249.↩︎
Karla N. Kendrick et al., “Plant-Based Diets and Risk of Hospitalization with Respiratory Infection: Results from the Atherosclerosis Risk in Communities (ARIC) Study,” Nutrients 15, no. 19 (October 5, 2023): 4265, https://doi.org/10.3390/nu15194265.↩︎
María Teresa García-Ordás et al., “Evaluation of Country Dietary Habits Using Machine Learning Techniques in Relation to Deaths from COVID-19,” Healthcare 8, no. 4 (September 29, 2020): 371, https://doi.org/10.3390/healthcare8040371.↩︎
Itandehui Castro-Quezada, Blanca Román-Viñas, and Lluís Serra-Majem, “The Mediterranean Diet and Nutritional Adequacy: A Review,” Nutrients 6, no. 1 (January 3, 2014): 231–48, https://doi.org/10.3390/nu6010231.↩︎
Rosa Casas, Emilio Sacanella, and Ramon Estruch, “The Immune Protective Effect of the Mediterranean Diet against Chronic Low-Grade Inflammatory Diseases,” Endocrine, Metabolic & Immune Disorders-Drug Targets 14, no. 4 (November 12, 2014): 245–54, https://doi.org/10.2174/1871530314666140922153350.↩︎
Ramon Estruch, “Anti-Inflammatory Effects of the Mediterranean Diet: The Experience of the PREDIMED Study,” Proceedings of the Nutrition Society 69, no. 3 (August 2010): 333–40, https://doi.org/10.1017/S0029665110001539.↩︎
Liselot Koelman, Caue Egea Rodrigues, and Krasimira Aleksandrova, “Effects of Dietary Patterns on Biomarkers of Inflammation and Immune Responses: A Systematic Review and Meta-Analysis of Randomized Controlled Trials,” Advances in Nutrition 13, no. 1 (January 2022): 101–15, https://doi.org/10.1093/advances/nmab086.↩︎
Hong-Yu Cheng et al., “Interactions Between the Gut Microbiota and the Host Innate Immune Response Against Pathogens,” Frontiers in Immunology 10 (March 29, 2019): 607, https://doi.org/10.3389/fimmu.2019.00607.↩︎
Anjali Verma et al., “Short-Chain Fatty Acid (SCFA) as a Connecting Link between Microbiota and Gut-Lung Axis─A Potential Therapeutic Intervention to Improve Lung Health,” ACS Omega 9, no. 13 (April 2, 2024): 14648–71, https://doi.org/10.1021/acsomega.3c05846.↩︎
Ji Yoo et al., “Gut Microbiota and Immune System Interactions,” Microorganisms 8, no. 10 (October 15, 2020): 1587, https://doi.org/10.3390/microorganisms8101587.↩︎
Hsin-Jung Wu and Eric Wu, “The Role of Gut Microbiota in Immune Homeostasis and Autoimmunity,” Gut Microbes 3, no. 1 (January 2012): 4–14, https://doi.org/10.4161/gmic.19320.↩︎
Wu and Wu.↩︎
Swadha Anand and Sharmila S. Mande, “Diet, Microbiota and Gut-Lung Connection,” Frontiers in Microbiology 9 (September 19, 2018): 2147, https://doi.org/10.3389/fmicb.2018.02147.↩︎
Wu and Wu, “The Role of Gut Microbiota in Immune Homeostasis and Autoimmunity.”↩︎
Wu and Wu.↩︎
Claire A. Woodall et al., “Respiratory Tract Infections and Gut Microbiome Modifications: A Systematic Review,” ed. Aran Singanayagam, PLOS ONE 17, no. 1 (January 13, 2022): e0262057, https://doi.org/10.1371/journal.pone.0262057.↩︎
Riddhi Trivedi and Kalyani Barve, “Gut Microbiome a Promising Target for Management of Respiratory Diseases,” Biochemical Journal 477, no. 14 (July 31, 2020): 2679–96, https://doi.org/10.1042/BCJ20200426.↩︎
Verma et al., “Short-Chain Fatty Acid (SCFA) as a Connecting Link between Microbiota and Gut-Lung Axis─A Potential Therapeutic Intervention to Improve Lung Health.”↩︎
Emanuele Rinninella et al., “What Is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases,” Microorganisms 7, no. 1 (January 10, 2019): 14, https://doi.org/10.3390/microorganisms7010014.↩︎
Emanuele Rinninella et al., “Food Components and Dietary Habits: Keys for a Healthy Gut Microbiota Composition,” Nutrients 11, no. 10 (October 7, 2019): 2393, https://doi.org/10.3390/nu11102393.↩︎
Yue Liu, Jiaqi Wang, and Changxin Wu, “Modulation of Gut Microbiota and Immune System by Probiotics, Pre-Biotics, and Post-Biotics,” Frontiers in Nutrition 8 (January 3, 2022): 634897, https://doi.org/10.3389/fnut.2021.634897.↩︎
Andrea Ballini et al., “About Functional Foods: The Probiotics and Prebiotics State of Art,” Antibiotics 12, no. 4 (March 23, 2023): 635, https://doi.org/10.3390/antibiotics12040635.↩︎
Liu, Wang, and Wu, “Modulation of Gut Microbiota and Immune System by Probiotics, Pre-Biotics, and Post-Biotics.”↩︎
Pengjun Zhou et al., “Unveiling the Therapeutic Symphony of Probiotics, Prebiotics, and Postbiotics in Gut-Immune Harmony,” Frontiers in Nutrition 11 (February 8, 2024): 1355542, https://doi.org/10.3389/fnut.2024.1355542.↩︎

