Report Author: Shenika Vilton, MD, MPH
Report Date: April 30, 2025
Table of Contents
Introduction
Polycystic Ovary Syndrome (PCOS) is a prevalent hormonal disorder that affects women of reproductive age, influencing not only their fertility but also their metabolic health and overall well-being. It is estimated that 6 to 13% of women worldwide are affected by PCOS, making it one of the most common endocrine disorders in women.1 The symptoms of PCOS are diverse, ranging from irregular menstrual cycles and acne to hirsutism (excess hair growth) and weight gain. These manifestations not only affect a woman’s physical appearance but also have profound impacts on her psychological health, fertility, and quality of life.2
The condition is often associated with significant health complications, including infertility, insulin resistance, and an increased risk of cardiovascular diseases. Insulin resistance, a core feature of PCOS, disrupts the body’s ability to properly utilize insulin, leading to elevated blood glucose levels and contributing to the development of type 2 diabetes. Furthermore, chronic inflammation plays a role in exacerbating the symptoms and underlying mechanisms of PCOS, making it crucial to understand the interconnectedness of these factors in managing the disorder effectively.3
Lifestyle interventions, such as dietary modifications and exercise, play a vital role in managing PCOS symptoms. Research supports the benefits of a low glycemic index (GI) diet, which helps regulate insulin levels and minimize inflammation.4 A diet rich in anti-inflammatory foods, including fruits, vegetables, and omega-3 fatty acids, can reduce systemic inflammation, while weight management through even modest weight loss has shown significant improvements in hormonal balance and insulin sensitivity.5 Additionally, regular physical activity is not only beneficial for managing body weight but also plays a critical role in improving insulin sensitivity, thereby alleviating some of the metabolic challenges faced by women with PCOS.6
Significance of the Topic
Polycystic Ovary Syndrome is a prevalent condition affecting women of reproductive age, with significant implications for both physical and mental health. While the immediate symptoms, such as irregular periods, acne, hirsutism, and weight gain, are widely recognized, the long-term effects of PCOS, particularly on metabolic health, are concerning.
A major concern is insulin resistance, which affects approximately 70% of women with PCOS, increasing their risk for type 2 diabetes.7 In the U.S., the prevalence of impaired glucose tolerance (31%-37%) and type 2 diabetes (7.5%-10%) in women with PCOS is notable.8 Insulin resistance also worsens hormonal imbalances, leading to symptoms like acne and excess hair growth. Managing insulin resistance is therefore crucial in preventing further metabolic complications.
Chronic low-grade inflammation, often linked to obesity, contributes to insulin resistance, dyslipidemia, and hypertension in women with PCOS.9 This combination significantly raises the risk of cardiovascular disease, as women with PCOS have a higher prevalence of high cholesterol, hypertension, and atherosclerosis.10
PCOS is also a leading cause of infertility, with 90-95% of women experiencing ovulation problems due to hormonal imbalances.11 Fertility-related issues are often the primary reason women seek medical help for PCOS, making it essential to explore effective treatments.
Psychologically, PCOS can contribute to anxiety, depression, and poor self-esteem, particularly due to visible symptoms like weight gain and excessive hair growth.12 The emotional strain of infertility and long-term health uncertainties further underscores the importance of addressing mental health in PCOS management.
Nutrition plays a key role in managing both the metabolic and hormonal aspects of PCOS. A low glycemic index (GI) diet, which includes whole grains, vegetables, and lean proteins while minimizing processed carbs, helps manage insulin levels and hormonal imbalances, improving symptoms like acne and hirsutism.13 Even modest weight loss can improve insulin sensitivity, regulate menstrual cycles, and restore ovulation.14
Additionally, a diet rich in anti-inflammatory foods, such as fruits, vegetables, nuts, seeds, and fatty fish, helps reduce chronic inflammation, regulate insulin sensitivity, and lower the risk of cardiovascular disease.15 This dietary approach can significantly benefit women with PCOS by addressing both the symptoms and underlying causes of the condition.
Pathology and Lifestyle Factors in PCOS
PCOS is a multifactorial endocrine disorder characterized by a complex interplay between genetic, hormonal, and environmental factors. The pathophysiology of PCOS involves a combination of hyperandrogenism, defined as the excessive presence of male sex hormones testostosterone, androsterone, androstenedione, dihydrotestoseterone (DHT) dehydroepiandodrosterone (DHEA) in women, insulin resistance, and chronic low-grade inflammation, which together contribute to the hallmark symptoms of the disorder: irregular menstrual cycles, anovulation, hirsutism, acne, and obesity.16
The defining feature of PCOS is hormonal imbalance, particularly an elevated level of androgens in women.17 Hyperandrogenism leads to several symptoms, including excess hair growth (hirsutism), acne, and scalp hair thinning. This imbalance is primarily due to abnormal ovarian function, where the ovaries produce excessive androgens, resulting in the disruption of normal ovulatory cycles.18 Normally, the hypothalamus-pituitary-ovarian axis regulates the release of hormones that trigger ovulation. In PCOS, however, this process is impaired, leading to anovulation, the lack or absence of ovulation, and the development of cysts in the ovaries.19
Another critical feature of PCOS is insulin resistance, which is observed in 50-70% of women with the condition.20 Insulin resistance occurs when the body’s cells become less responsive to insulin, a hormone responsible for regulating blood sugar levels.21 In response, the pancreas compensates by producing more insulin.22 This increased insulin level can promote excessive androgen production in the ovaries and exacerbate the hormonal imbalances in PCOS. Insulin resistance is also strongly associated with metabolic disturbances, such as obesity, dyslipidemia, and hypertension, which contribute to the increased risk of developing type 2 diabetes and cardiovascular disease in women with PCOS.23
Chronic low-grade inflammation, a persistent low level inflammatory state occurring in the absence of infection or injury, is another pathological factor in PCOS. Elevated levels of pro-inflammatory cytokines, such as C-reactive protein (CRP), have been observed in women with PCOS, particularly those who are overweight or obese.24 This inflammation contributes to insulin resistance and the development of other metabolic abnormalities, creating a vicious cycle that exacerbates the symptoms and long-term health risks associated with PCOS.
Lifestyle factors, particularly diet and physical activity, play a crucial role in modulating the severity and progression of PCOS. Diets rich in high glycemic index (GI) foods, which lead to rapid spikes in blood sugar and insulin levels, can worsen insulin resistance and contribute to weight gain. Conversely, a low-GI diet, rich in fiber, lean proteins, and whole grains, has been shown to improve insulin sensitivity and hormonal balance in women with PCOS.25 Moreover, anti-inflammatory foods such as fruits, vegetables, nuts, seeds, and omega-3 fatty acids found in fatty fish help reduce inflammation and regulate insulin levels, further improving metabolic outcomes in PCOS.26
Exercise is another essential lifestyle intervention. Regular physical activity has been shown to reduce insulin resistance, improve metabolic health, and promote weight loss in women with PCOS. Even moderate exercise, such as walking or cycling, can enhance insulin sensitivity and improve reproductive function by reducing the production of excess androgens.27 Exercise also improves cardiovascular health, which is particularly important for women with PCOS who are at higher risk for heart disease.28
The connection between PCOS and overall health extends beyond the reproductive system. Women with PCOS face a significantly higher risk of developing type 2 diabetes, with studies indicating that 31-37% of women with PCOS have impaired glucose tolerance and 7.5-10% have type 2 diabetes.29 In addition, cardiovascular risk factors such as high blood pressure, elevated cholesterol levels, and atherosclerosis are more prevalent in women with PCOS.30 These metabolic disturbances highlight the importance of early detection and proactive management of PCOS to reduce long-term health risks.
Key Nutrients and Food
Polycystic ovarian syndrome is a hormonal condition that can impact insulin sensitivity, weight management, and reproductive health. It requires a diet rich in key nutrients to help manage symptoms and improve overall well-being. Foods high in fiber, such as whole grains, vegetables, and legumes, can help stabilize blood sugar levels and manage insulin resistance.31 Healthy fats from sources like avocados, nuts, seeds, and fatty fish (like salmon) support hormone regulation and reduce inflammation.32 Lean proteins, including poultry, tofu, and beans, help maintain muscle mass and support metabolic function.33 Additionally, a diet with low glycemic index (GI) foods, such as leafy greens, berries, and sweet potatoes, can prevent blood sugar spikes.34 Consuming anti-inflammatory foods like turmeric, cinnamon, and omega-3-rich foods can also help manage the symptoms of PCOS.35 Some women may benefit from incorporating supplements like inositol, magnesium, or vitamin D for metabolic and hormonal balance.36 This literature review explores the impacts of these nutrients and the diets that incorporate them into management of PCOS.
Specific Nutrient: Fiber
What Is It
Dietary fiber, also known as roughage, is the indigestible portion of plant-derived foods that cannot be completely broken down by human digestive enzymes.37 It consists of non-starch polysaccharides and other plant components such as cellulose, resistant starch, resistant dextrins, inulins, lignins, chitins, pectins, beta-glucans, and oligosaccharides.38 Fiber is typically categorized into two main types: soluble and insoluble. Soluble fiber attracts water and forms a gel-like substance in the gut, which can slow down digestion, helping to control blood sugar levels and reduce cholesterol.39 On the other hand, insoluble fiber adds bulk to stool and helps food pass more quickly through the stomach and intestines, promoting regularity and preventing constipation.40
How It Works
Dietary fiber plays a crucial role in managing symptoms of polycystic ovarian syndrome, although it does not cure the condition itself. A high-fiber diet has been shown to alleviate several PCOS-related issues, including insulin resistance, hyperandrogenism (excess male hormones like testosterone), and metabolic inflammation.41 By improving insulin sensitivity and blood sugar control, fiber intake can help regulate blood sugar levels and potentially reduce testosterone and DHEA production (key hormones in the testosterone pathway, produced excessively in polycystic ovarian syndrome).42 Additionally, increased fiber consumption supports gut health, which may improve mood and reduce food cravings commonly experienced by women with PCOS.43 Fiber also promotes satiety, helping with weight management, which can be particularly beneficial for those with PCOS who struggle with obesity.44
Dietary fiber works through several mechanisms in the body, primarily bulking, viscosity, and fermentation. Insoluble fibers, like wheat bran and cellulose, absorb water and increase stool bulk, promoting regular bowel movements.45 Soluble(viscous) fibers, such as beta-glucan and psyllium, form a gel-like substance in the gut, increasing the viscosity of intestinal contents.46 This slows digestion and nutrient absorption, which can help lower blood cholesterol levels and reduce the glycemic response (the rise in blood sugar after eating) in individuals with PCOS. The increased viscosity also enhances feelings of fullness, aiding in weight management.47 The effectiveness of viscous fibers in reducing nutrient absorption varies depending on their level of viscosity, making some fibers more beneficial than others in this regard.
Some dietary fibers also serve as food for beneficial gut bacteria in the large intestine, a process known as fermentation.48 This process produces short-chain fatty acids (SCFAs) like acetate, propionate, and butyrate. These SCFAs have several important effects, including lowering intestinal pH, which increases mineral absorption, providing energy for colon cells, stimulating the production of hormones that improve satiety and insulin sensitivity, and supporting a healthy immune system.49 Different fibers ferment to varying degrees and produce different ratios of SCFAs, leading to diverse health effects. For example, oligosaccharides and resistant starch are fully fermented, while polydextrose and resistant maltodextrin are partially fermented.50 Through these processes, dietary fiber contributes to gut health, helps regulate blood sugar and cholesterol levels, and supports weight management in PCOS patients.
While these effects can significantly improve the quality of life for women with PCOS, it is important to recognize that dietary fiber is a tool for managing the condition, not a cure for the underlying hormonal imbalances. Incorporating a high-fiber diet, alongside other lifestyle modifications and medical treatments, can be an effective approach to managing PCOS symptoms and improving overall health.
What the Research Is Telling Us
Dietary fiber has become an essential factor in managing polycystic ovary syndrome. Increasing fiber intake through whole grains, fruits, vegetables, and legumes may help alleviate PCOS symptoms. A cohort study compared the dietary habits, physical activity levels, metabolic profiles, and hormonal markers of women with PCOS to healthy controls.51 The study found that women with PCOS consumed significantly less dietary fiber (median: 19.6 g vs. 24.7 g) and magnesium (median: 238.9 mg vs. 273.9 mg) than the control group, despite similar caloric intake and physical activity levels. Among women with PCOS, those with insulin resistance had even lower fiber intake (median: 18.2 g vs. 22.1 g) compared to those without insulin resistance.
The study revealed negative correlations between fiber intake and insulin resistance-related markers, such as fasting insulin, glucose tolerance, testosterone levels, and dehydroepiandrosterone sulfate (DHEAS). Notably, fiber intake and body mass index (BMI) together accounted for 54% of the observed variance in insulin resistance in women with PCOS. The findings suggest that inadequate fiber consumption may exacerbate insulin resistance and hyperandrogenism, worsening the metabolic and hormonal imbalances associated with PCOS. The authors propose that increasing dietary fiber and magnesium intake could help manage PCOS symptoms by improving insulin sensitivity and reducing androgen levels.
However, the study also has limitations, such as its reliance on self-reported dietary records and its observational nature, which prevents establishing causality. Future research should focus on randomized controlled trials to confirm whether increasing fiber intake directly improves metabolic outcomes in women with PCOS.
A high-fiber diet has been shown to improve chronic metabolic inflammation, reproductive function, and brain-gut peptide secretion in PCOS patients. In a clinical trial examining the role of gut microbiota in the diverse clinical manifestations of PCOS, including metabolic and hormonal imbalances, 25 women with PCOS were randomly assigned to two groups: one group (n = 14) received a high-fiber diet called the WTP diet (comprising whole grains, traditional Chinese medicinal foods, and prebiotics), and the other group (n = 11) received the WTP diet combined with acarbose, an alpha-glucosidase inhibitor antidiabetic medication used to lower blood sugars and prevent the breakdown of starch into sugars.52 The intervention lasted 12 weeks, during which various parameters were assessed, including sex hormones, glycolipid metabolism markers, inflammatory factors, brain-gut peptides, and gut microbiota composition.
Both interventions improved PCOS-related clinical phenotypes, reduced inflammation, and regulated brain-gut peptide secretion. Analysis of gut microbiota diversity showed significant changes in both groups. Beneficial bacterial groups, such as Bifidobacterium and Lactobacillus, were enriched, while potentially harmful bacteria like Bacteroides vulgatus, Alistipes, Blautia, and Roseburia were inhibited. These changes in gut microbiota were strongly correlated with improvements in key clinical markers such as testosterone levels, insulin resistance (measured by HOMA-IR), inflammatory markers (e.g., α-1-acid glycoprotein), and leptin levels. Conversely, positive correlations were observed between beneficial bacterial groups and adiponectin and spexin levels, both associated with improved metabolic health.
The study concluded that a high-fiber diet alone can alleviate PCOS symptoms by reducing chronic inflammation and regulating brain-gut peptides. The remodeling of gut microbiota appeared to play a central role in these improvements. These findings suggest that dietary interventions targeting gut microbiota could be a promising approach for managing PCOS. However, the authors emphasized the need for larger clinical trials to confirm these results and further investigate the mechanisms underlying the gut microbiota’s role in PCOS pathology.
Recent research indicates that women with PCOS often have lower fiber intake compared to healthy controls, which may worsen their symptoms and metabolic complications. A meta-analysis aimed at determining whether women with PCOS consume less fiber than those without the condition found that women with PCOS indeed consumed significantly less dietary fiber.53 The analysis of 12 studies revealed that PCOS patients consumed significantly less dietary fiber than controls, despite similar total energy intake. Specifically, women with PCOS had a standardized mean difference of -0.32 (95% CI: -0.50, -0.14, p < 0.001) in dietary fiber intake compared to controls.54 This fiber deficiency is concerning, as adequate fiber consumption is associated with improved insulin sensitivity, reduced inflammation, and better hormonal balance. Lower fiber intake may contribute to metabolic challenges associated with PCOS, such as insulin resistance and obesity. The authors hypothesized that lifestyle factors, dietary habits, or cultural influences may explain this difference in fiber consumption. This underscores the importance of addressing dietary patterns in women with PCOS and suggests that increasing fiber intake could be a valuable part of nutritional interventions for this population.
While more research is needed to establish optimal fiber intake levels for PCOS management, current evidence strongly supports the inclusion of fiber-rich foods in the diets of women with PCOS. Nutritional strategies focusing on increased fiber consumption may offer a promising, non-pharmacological approach to alleviating PCOS symptoms and improving overall health outcomes in affected women.
Dosage Recommendation Based on Research
PCOS individuals are recommended to take up to two times the recommended intake of fiber as individuals without PCOS, aiming for 30-35g of fiber per day. The general recommendation of 14g of fiber per 1000 kilocalories consumed may be insufficient for women with PCOS.55
Table 1 Dosage Recommendation for Fiber
| What’s the Daily Recommended Intake? | How much do I take? | Don’t take more than… | When will I see results? |
|---|---|---|---|
| 14g per day | 30-35g per day | 70g per day | 2-4 weeks minimum |
Facts and Misconceptions
A common misconception is that fiber is only important for digestive health. While fiber does play a crucial role in digestion, its benefits extend to various aspects of overall health. Studies have demonstrated that fiber intake can impact blood sugar control, weight management, and even cardiovascular health. For instance, high-viscosity, gel-forming fibers have been shown to improve glycemic control and lower cholesterol levels in a dose-dependent manner.56
Some people believe that all fiber supplements will lead to weight loss. However, research indicates that the effectiveness of fiber supplements for weight management can vary. A study comparing different types of fiber supplements found that a viscous, gel-forming, nonfermented fiber (psyllium) was more effective for sustained weight loss compared to a less viscous, readily fermented fiber (partially hydrolyzed guar gum).57
There is also a misconception that increasing fiber intake always leads to improved health outcomes. While fiber is generally beneficial, individual responses to fiber supplements can vary significantly. A Stanford Medicine study revealed that some people may experience negative effects from certain types of fiber supplements. For example, high doses of inulin caused inflammation in some individuals, while others responded well to it.58
Lastly, many people believe that the source of fiber does not matter as long as they meet their daily intake requirements. However, research suggests that a diverse range of fiber sources may be more beneficial than relying on a single type. Different fibers can have varying effects on the gut microbiome and metabolic health, and a combination of soluble and insoluble fibers from various sources may provide the most comprehensive health benefits.59
Food Sources
Dietary fiber has many sources with differing quantities per food item. High-fiber foods include legumes, whole grains, fruits, vegetables, nuts and seeds.60
· Legumes:
o Navy beans: 19g of fiber per cup
o Lentils: 16g of fiber per cup
o Pinto beans: 15g of fiber per cup
o Black beans: 15g of fiber per cup
o Chickpeas: 12g of fiber per cup
· Whole grains:
o Oats: 16.5g of fiber per cup
o Quinoa: 10.0g of fiber per cup
o Brown rice: 3.5g of fiber per cup (cooked)
o Whole wheat flour: 14.6g of fiber per cup
o Barley: 6.0g of fiber per cup (cooked)
· Fruits:
o Raspberries: 8g of fiber per cup
o Pears: 5.5g of fiber per medium fruit
o Apples: 4.4g of fiber per medium fruit
o Bananas: 3.1g of fiber per medium fruit
o Oranges: 3.1g of fiber per medium fruit
· Vegetables:
o Artichokes: 6.9g of fiber per medium artichoke
o Broccoli: 5.1g of fiber per cup
o Carrots: 3.1g of fiber per cup
o Collard greens: 7.6g of fiber per cup
o Swiss chard: 3.7g of fiber per cup
· Nuts and seeds:
o Chia seeds: 10g of fiber per ounce
o Flax seeds: 8g of fiber per ounce
o Almonds: 3.5g of fiber per ounce
o Pistachios: 3g of fiber per ounce
o Sunflower seeds: 3g of fiber per ounce
Supplemental Sources
Fiber supplements are available in diverse forms to accommodate different preferences and needs, including powders, capsules, tablets, gummies, wafers, and bars. These supplements can be particularly beneficial for individuals who struggle to meet their daily fiber requirements through diet alone, offering a convenient way to increase fiber intake. Common ingredients in fiber supplements include psyllium, inulin, wheat dextrin, acacia gum, and methylcellulose, each providing unique benefits and characteristics. While whole foods remain the preferred source of fiber due to their additional nutritional benefits, supplements can serve as a valuable tool to bridge nutritional gaps and support digestive health, blood sugar management, and heart health for individuals with PCOS.61
Side Effects
Common side effects of excessive fiber intake include bloating, gas, and abdominal discomfort. Fiber can interact with certain medications by affecting their absorption, so it is important to consult with a healthcare provider.
Bottom Line
The strength of recommendation for increasing fiber intake is high, given the substantial evidence supporting its various health benefits. However, individual needs may vary, and it is important to increase fiber intake gradually and consume adequate fluids. While supplements can be beneficial, obtaining fiber from a variety of whole food sources is generally recommended for optimal health benefits.
Specific Nutrient: Omega-3
What Is It
Omega-3 polyunsaturated fatty acids (PUFAs) are essential fats that the human body cannot produce on its own. They are found naturally in certain fish, plant oils, and nuts. The three main types are alpha-linolenic acid (ALA), eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA).62 Omega-3 fatty acids are being investigated for their potential role in managing polycystic ovary syndrome (PCOS) due to their anti-inflammatory properties and potential to improve metabolic parameters, and are considered quintessential healthy fats.63
How It Works
Omega-3 fatty acids are increasingly being used as a complementary approach in managing symptoms associated with PCOS. These essential fats have shown promise in alleviating several PCOS-related symptoms and metabolic disturbances. Omega-3 supplementation has been found to improve insulin resistance, helping regulate blood sugar levels and reduce the risk of type 2 diabetes.64 It can also help lower inflammation, decrease total cholesterol(decreasing LDL and HDL and increasing PON-1) and triglyceride levels, and potentially aid in regulating menstrual cycles.65 Omega-3 may also contribute to balancing hormone levels, particularly by reducing testosterone, which could help alleviate symptoms like acne, excessive hair growth, and hair loss.66 While these benefits are encouraging, it is important to note that omega-3 is not a standalone treatment for PCOS and should be used as part of a comprehensive management plan.
Omega-3 fatty acids exert their effects through multiple mechanisms in the human body, particularly in relation to inflammation and metabolism. These essential fatty acids are incorporated into cell membranes, influencing cell signaling and gene expression, and serve as precursors for anti-inflammatory molecules.67 In terms of inflammation, omega-3 fatty acids, especially EPA and DHA, compete with omega-6 fatty acids for enzymes involved in eicosanoid production, the creation of lipid-based signaling molecules that regulate inflammatory responses. This competition leads to the formation of less inflammatory and even anti-inflammatory mediators, many of which can counter the inflammatory processes and symptoms seen in PCOS.68 Omega-3s inhibit the enzyme cyclooxygenase (COX), which is responsible for producing pro-inflammatory hormones.69 Additionally, EPA and DHA give rise to specialized pro-resolving mediators (SPMs) such as resolvins, protectins, and maresins, which actively resolve inflammation and promote tissue homeostasis.70
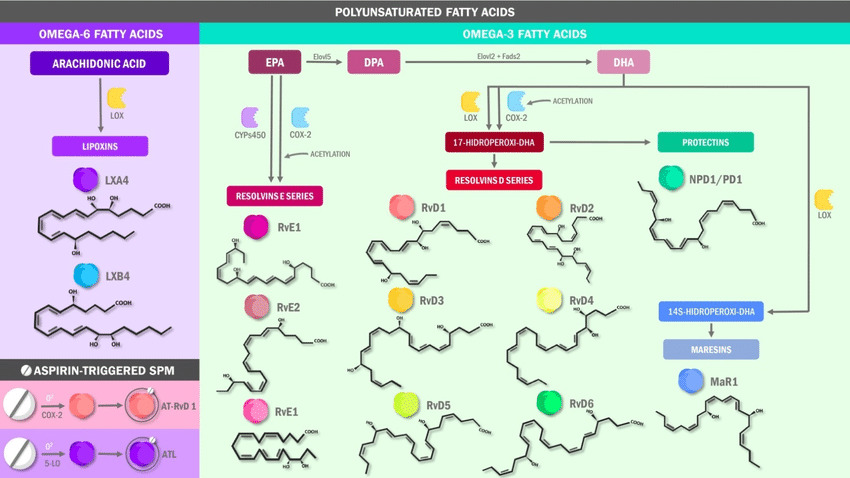
Figure 1 Metabolism of specialized pro-resolving lipid mediators, source: DOI: 10.3390/ijms22191037
Metabolically, omega-3 fatty acids play a crucial role in improving insulin sensitivity in polycystic ovarian syndrome. They activate peroxisome proliferator-activated receptors (PPARs), particularly PPAR-gamma, which regulates inflammatory gene expression and NFκB activation.71 This activation can lead to improved insulin signaling and glucose uptake in cells. Omega-3s also inhibit the sterol regulatory element-binding protein 1 (SREBP-1), which is involved in lipogenesis, potentially reducing triglyceride synthesis in the liver.72
The effects of omega-3 fatty acids on the endocrine system are also noteworthy. They may help regulate hormone production and signaling, potentially influencing menstrual cycle regularity in women with PCOS.73 Additionally, omega-3s have been shown to reduce the levels of androgens, which are often elevated in PCOS and contribute to many of its symptoms.74
What the Research Is Telling Us
Research on omega-3 fatty acids for managing PCOS is promising, though still evolving. A randomized, double-blind, placebo-controlled clinical trial with 61 participants found that omega-3 supplementation significantly reduced both the total cholesterol/HDL-cholesterol and LDL-cholesterol/HDL-cholesterol ratios.75 It also increased serum paraoxonase 1 (PON1) activity compared to a placebo. Participants were divided into two groups: the omega-3 group, which received 4 grams of omega-3 fatty acids per day for 8 weeks, and the placebo group, which received a placebo for the same period. Fasting blood samples, anthropometric measurements, and dietary intake data were collected at both baseline and after the intervention. Lipid parameters including total cholesterol (TC), triglycerides (TG), HDL-C, LDL-C, and the TC/HDL-C and LDL-C/HDL-C ratios were measured. Serum PON1 activity was assessed through arylesterase activity.
The results showed significant improvements in the omega-3 group. Specifically, the TC/HDL-C and LDL-C/HDL-C ratios decreased significantly, indicating a favorable effect on cardiovascular risk factors. Additionally, serum PON1 activity increased, suggesting an improvement in antioxidant defense mechanisms. However, no significant differences were found in serum TG or HDL-C levels between the groups. These findings suggest that omega-3 fatty acids may help reduce cardiovascular disease risk in PCOS patients by improving serum PON1 activity and lowering TC/HDL-C and LDL-C/HDL-C ratios. This study highlights omega-3 supplementation as a potential strategy for reducing cardiometabolic risks in women with PCOS. However, there are some limitations. The study focused exclusively on overweight or obese PCOS patients, which limits its applicability to individuals with other body mass index (BMI) categories. Additionally, other antioxidant enzymes were not measured, which could have provided a more comprehensive assessment of oxidative stress. Despite these limitations, the study offers valuable insights into the potential benefits of omega-3 fatty acids for managing metabolic complications associated with PCOS.
A systematic review and meta-analysis of nine trials involving 591 participants found that omega-3 supplementation may improve insulin resistance (as measured by the homeostatic model assessment, or HOMA), decrease total cholesterol and triglyceride levels, and increase adiponectin levels in women with PCOS.76 The researchers conducted an extensive search of multiple databases to identify relevant randomized controlled trials (RCTs). These studies examined the effects of omega-3 supplementation on hormonal profiles, lipid profiles, glucose metabolism, menstrual cycle regulation, and ovarian morphology. Data from the selected studies were extracted and analyzed using meta-analytic techniques, and the quality of the studies was assessed to evaluate the reliability of the findings.
The meta-analysis revealed several beneficial effects of omega-3 supplementation in women with PCOS. These included improved lipid profiles (significant reductions in triglyceride levels and increases in HDL-cholesterol levels), hormonal regulation (reduced testosterone levels and increased sex hormone-binding globulin levels), and improved glucose metabolism (enhanced insulin sensitivity and reduced fasting glucose levels). Some studies also suggested improvements in menstrual cycle regularity and ovarian morphology, although these results were less consistent across the studies.
Based on these findings, the study concluded that omega-3 fatty acid supplementation has beneficial effects on various aspects of PCOS, including lipid profiles, hormonal balance, and glucose metabolism. The authors suggest that omega-3 fatty acids could serve as an adjunctive therapy for managing PCOS. However, they also acknowledge limitations, such as variability in the dosages, study durations, and participant characteristics across the studies. While these results are encouraging, the authors call for larger, long-term studies to establish definitive recommendations for omega-3 supplementation in PCOS management.
Dosage Recommendation Based on Research
There is no specific FDA recommendation for omega-3 intake for PCOS, but the American Heart Association recommends 1-2 servings of fatty fish per week for general health. In studies on PCOS, dosages have ranged from 1-4 g per day of combined EPA and DHA.77 Based on current research, a range of 1-3 g per day of combined EPA and DHA may be beneficial for women with PCOS, but individual needs may vary.78
Table 2 Dosage Recommendation for Omega-3
| What’s the Daily Recommended Intake? | How much do I take? | Don’t take more than… | When will I see results? |
|---|---|---|---|
| 1.1-1.6 g ALA for women, 1.6 g ALA for men | 1-3 g combined EPA and DHA per day | 3g per day | 8-12 weeks minimum |
Facts and Misconceptions
It is a fact that omega-3s can help improve metabolic parameters in PCOS, but a misconception that omega-3s alone can cure PCOS.79 Both plant-based (ALA) and marine-based (EPA and DHA) omega-3s have health benefits, contrary to the misconception that only fish oil provides beneficial omega-3s.80
Food Sources
Fatty fish like salmon, mackerel, sardines, and tuna are rich in omega-3s. Plant-based sources include flaxseeds and flaxseed oil, chia seeds, walnuts, canola oil, and soybean oil.81 These foods offer a natural way to increase omega-3 intake.
Table 3 Food Sources for Omega-3
| Food (serving size) | ALA (g) | DHA (g) | EPA (g) |
|---|---|---|---|
| Flaxseed oil (1 tbsp) | 7.26 | – | – |
| Chia seeds (1 oz) | 5.06 | – | – |
| English walnuts (1 oz) | 2.57 | – | – |
| Flaxseed, whole (1 tbsp) | 2.35 | – | – |
| Salmon, Atlantic, farmed (3 oz) | – | 1.24 | 0.59 |
| Salmon, Atlantic, wild (3 oz) | – | 1.22 | 0.35 |
| Herring, Atlantic (3 oz) | – | 0.94 | 0.77 |
| Canola oil (1 tbsp) | 1.28 | – | – |
| Sardines, canned (3 oz) | – | 0.74 | 0.45 |
| Mackerel, Atlantic (3 oz) | – | 0.59 | 0.43 |
| Salmon, pink, canned (3 oz) | 0.04 | 0.63 | 0.28 |
| Soybean oil (1 tbsp) | 0.92 | – | – |
| Trout, rainbow, wild (3 oz) | – | 0.44 | 0.40 |
Supplemental Sources
Omega-3 supplements are available in various forms, including fish oil capsules, krill oil, and algae-based supplements.82 Fish oil capsules are the most common and well-studied form. While supplements can provide concentrated doses of EPA and DHA, obtaining omega-3s from food sources is generally preferable as whole foods offer additional nutrients and fiber.
Side Effects
Common side effects of omega-3 supplements include a fishy aftertaste, gastrointestinal discomfort, and nausea.83 They may interact with blood thinners, such as warfarin, and increase bleeding risk.84 Individuals with fish allergies should avoid fish oil supplements and opt for algae-based alternatives.
Bottom Line
The strength of recommendation for using omega-3s in PCOS management is moderate. While research shows promising results, particularly in improving metabolic parameters and reducing inflammation, more large-scale, long-term studies are needed to establish definitive recommendations. Incorporating omega-3-rich foods into the diet is a safe and potentially beneficial strategy for women with PCOS, but supplementation should be discussed with a healthcare provider to determine individual needs and potential risks.
Specific Nutrient: Curcumin
What Is It
Curcumin, more commonly known as Turmeric (Curcuma longa), is a flowering plant in the ginger family Zingiberaceae, native to the Indian subcontinent and Southeast Asia.85 It has been used for thousands of years in traditional medicine and as a culinary spice, particularly in India, China, and Southeast Asia. The plant has been used to treat a multitude of disorders and chronic conditions such as digestive disorders, rheumatoid arthritis, conjunctivitis, liver ailment, urinary tract infection, smallpox, chickenpox, wounds and regulation of menstruation.86 The main active compound in turmeric is curcumin, a bright yellow chemical that gives turmeric its characteristic color.87 Curcumins role as an antioxidant and anti-inflammatory substance, makes turmeric’s use in regulating chronic conditions such as polycystic ovarian syndrome appealing.88
How It Works
Curcumin, the primary bioactive compound in turmeric, exerts its beneficial effects through multiple mechanisms, primarily focusing on anti-inflammatory, antioxidant, and metabolic pathways all of which exerts high effects on the pathogenesis of PCOS. Its anti-inflammatory properties are mediated through the modulation of key signaling pathways, including the suppression of nuclear factor kappa-B (NF-κB) activation, modulation of Mitogen-activated protein kinases (MAPK) and Activator Protein 1 (AP-1) pathways, and upregulation of Peroxisome proliferator-activated receptor gamma (PPAR-γ).89 These actions lead to a decrease in pro-inflammatory gene expression and cytokine production.
The antioxidant properties of curcumin stem from its ability to directly neutralize free radicals and enhance the body’s antioxidant defenses. It scavenges various forms of reactive oxygen and nitrogen species, enhances the activity of antioxidant enzymes, and activates Nuclear factor erythroid 2-related factor 2 (Nrf2), a key regulator of antioxidant protein expression.90 Additionally, curcumin’s metal chelation properties, particularly for iron and copper, contribute to its antioxidant effects by reducing the generation of reactive oxygen species.
Furthermore, curcumin demonstrates significant metabolic benefits, particularly in glucose homeostasis and insulin sensitivity. It enhances insulin sensitivity by upregulating glucose transporter genes, activating AMP kinase, and promoting PPAR ligand-binding activity.91 Curcumin also inhibits hepatic glucose production and reduces insulin resistance through its anti-inflammatory and antioxidant actions.92 These diverse mechanisms collectively contribute to curcumin’s potential therapeutic effects in various inflammatory, oxidative, and metabolic conditions.
What the Research Is Telling Us
Recent research has highlighted the potential therapeutic effects of curcumin for polycystic ovary syndrome (PCOS). Several studies have demonstrated curcumin’s ability to alleviate various symptoms and address underlying mechanisms of PCOS.
A 2024 study published in Frontiers in Endocrinology provides a comprehensive investigation into curcumin’s therapeutic potential for PCOS using a rat model.93 The researchers induced PCOS in rats through daily subcutaneous injections of dehydroepiandrosterone (DHEA) at a dose of 60 mg/kg for 21 days. Curcumin was then administered at a daily dose of 50 mg/kg for another 21 days. The study’s strength lies in its multifaceted approach, examining hormonal regulation, ovarian function, oxidative stress markers, and the expression of peroxisome proliferator-activated receptor gamma (PPAR-γ). The results were statistically significant and promising across multiple parameters. Curcumin treatment led to substantial reductions in key hormones, including estradiol (p = 0.02), luteinizing hormone (LH) (p = 0.009), testosterone (p = 0.003), and the LH/FSH ratio (p = 0.008). Additionally, improvements in ovarian morphology and function were observed, such as the normalization of estrous cycles and a decrease in follicular cysts. Oxidative stress markers like reactive oxygen species (p = 0.005) showed significant reductions, while antioxidant levels such as superoxide dismutase (p = 0.04), glutathione peroxidase (p = 0.002), and glutathione (p = 0.02) increased. A key finding was the significant upregulation of PPAR-γ expression in ovarian tissues following curcumin treatment, which the authors suggest may be a crucial mechanism behind curcumin’s therapeutic effects in regards to PCOS’s high inflammation process. The study also proposes that curcumin’s efficacy may be mediated through its antioxidant properties, PPAR-γ upregulation, and potentially by inhibiting the NF-κB signaling pathway, although this was not directly measured in the study. While the research offers compelling evidence for curcumin’s potential in PCOS treatment, it acknowledges limitations, such as the exclusive use of a rat model, and calls for further research to better understand the mechanisms behind PPAR-γ upregulation and to determine optimal dosing for human subjects.
Another experimental animal study published in Nature in 2024 revealed that curcumin mitigates PCOS in mice by inhibiting the TLR4/MyD88/NF-κB signaling pathway and reducing intestinal mucosal permeability.94 The study used C57BL/6J mice, divided into four groups: control, control with curcumin, PCOS model, and PCOS model with curcumin treatment. PCOS was induced in the model groups through DHEA injections, and curcumin was administered for 45 days. The findings showed significant improvements in hormonal regulation, ovarian function, and intestinal barrier integrity. Curcumin treatment reduced testosterone and luteinizing hormone levels, lowered the LH/FSH ratio, increased estradiol levels, and promoted the formation of tight junction proteins in colon tissues. These results indicate the potential management of hyperandrogenism and the symptoms that result in PCOS. Ovarian tissue also showed improvements, with a reduction in cystic follicles and increased corpus luteum formation showing improvement in menstrual regulation. Furthermore, curcumin treatment led to decreased levels of inflammatory markers and an increase in anti-inflammatory IL-10. The study concluded that curcumin’s therapeutic effects on PCOS are mediated through the inhibition of the TLR4/MyD88/NF-κB signaling pathway and the reduction of intestinal mucosal permeability.
Clinical trials have also shown promising results. A randomized, placebo-controlled trial published in Progress in Nutrition in 2021 reported that curcumin supplementation improved anthropometric measurements and glycemic parameters in women with PCOS, though it did not significantly affect hormone or lipid profiles.95 The study involved 30 participants diagnosed with PCOS, randomly assigned to either a curcumin or placebo group. The curcumin group received 93.34 mg of curcumin daily for eight weeks. After the intervention, the curcumin group showed significant improvements in anthropometric measurements, such as a mean weight loss of 5.8 ± 2.3 kg, reduced body fat mass, and a reduction in waist circumference (7.2 ± 3.5 cm). Glycemic parameters, including fasting blood glucose, fasting insulin, and HOMA-IR, also improved significantly in the curcumin group. Additionally, C-reactive protein (CRP) levels were lower in the curcumin group, suggesting an anti-inflammatory effect. However, no significant differences were found in lipid parameters or hormone levels between the two groups. The researchers concluded that curcumin supplementation improved anthropometric and glycemic parameters in women with PCOS, although it did not significantly affect hormone or lipid profiles.
Curcumin’s anti-inflammatory and antioxidant properties appear to be central to its therapeutic effects in PCOS. An animal study showed that curcumin significantly reduced several inflammatory markers in both plasma and ovarian tissue of PCOS mice.96 Pro-inflammatory cytokines such as IL-17A, IL-6, and TNF-α decreased, while the anti-inflammatory cytokine IL-10 increased. The study also found significant changes in the TLR4/MyD88/NF-κB signaling pathway, with increased levels of NF-κB p65, TLR-4, and MyD88 in ovarian tissues of curcumin-treated PCOS mice. These results suggest that curcumin may reduce pro-inflammatory cytokines while increasing anti-inflammatory ones. Coupled with its ability to modulate insulin sensitivity and reduce oxidative stress, curcumin shows promise as a therapeutic option for PCOS management.
A meta-analysis found that daily intake of 500-1500 mg of curcumin reduced fasting insulin levels and enhanced cellular insulin sensitivity in PCOS patients.97 This study, a systematic review and meta-analysis of randomized controlled trials (RCTs), assessed curcumin’s effects on glycemic control and lipid profiles in women with PCOS found similar results. Researchers examined five electronic databases (PubMed, Embase, Scopus, Web of Science, and Cochrane Library) up to November 2020, identifying three RCTs with a total of 168 participants that met the inclusion criteria. The primary aim was to evaluate curcumin’s impact on glycemic control, measured by fasting glucose, fasting insulin, Homeostasis Model Assessment of Insulin Resistance (HOMA-IR), and the quantitative insulin sensitivity check index (QUICKI). Secondary goals included assessing curcumin’s effect on lipid profiles, including high-density lipoprotein (HDL), low-density lipoprotein (LDL), triglycerides, and total cholesterol. The meta-analysis revealed that curcumin significantly improved fasting glucose, fasting insulin, HOMA-IR, QUICKI, and HDL levels, as well as total cholesterol. However, curcumin had no significant effect on LDL or triglyceride levels. Trial sequential analysis confirmed the robustness of the findings for fasting glucose, fasting insulin, HOMA-IR, QUICKI, and total cholesterol. The authors concluded that curcumin may improve glycemic control and lipid metabolism in PCOS patients without significant adverse effects. They suggested that further studies are needed to explore curcumin’s effects on hyperandrogenism in PCOS patients.
Despite these encouraging findings, more large-scale clinical trials are necessary to fully establish curcumin’s efficacy and optimal dosing for PCOS treatment. However, the current body of research suggests that curcumin holds significant potential as a complementary therapy for managing PCOS symptoms and underlying metabolic disturbances.
Dosage Recommendation Based on Research
The FDA and NIH have not established a daily recommended intake for turmeric or curcumin. However, based on research, doses of 500-2000 mg of turmeric extract per day, standardized to 95% curcuminoids, have been used in various studies.98 It is important not to exceed 2000 mg per day to avoid potential side effects.99 Results may be observed after 1-4 weeks of consistent use, but this can vary depending on the individual and the specific condition being addressed.
Table 4 Dosage Recommendation for Curcumin
| What’s the Daily Recommended Intake? | How much do I take? | Don’t take more than… | When will I see results? |
|---|---|---|---|
| 500mg-1000mg per day | 500mg-1000mg per day | 2000mg per day | 1 to 4 weeks |
Facts and Misconceptions
A well-established fact about curcumin is its poor bioavailability when consumed orally. This limitation is due to its rapid metabolism in the liver and intestinal wall, as well as its quick elimination from the body.100 However, this fact has led to the development of various formulations and combinations aimed at enhancing curcumin’s absorption and effectiveness. For instance, the addition of piperine, a compound found in black pepper, has been shown to increase curcumin’s bioavailability by up to 2000%.101
Food Sources
Curcumin, the primary bioactive compound in turmeric, is not commonly found in significant amounts in foods other than turmeric itself. While turmeric remains the principal dietary source of curcumin, small quantities may be present in various turmeric-flavored or turmeric-containing foods. These sources include:
· Fresh turmeric root
· Dried, ground turmeric powder
· Turmeric tea
· Curry powder (which contains turmeric)
Additionally, curcumin can be found in small amounts in other culinary applications and products:
· Curries and curry-based sauces, particularly in Indian, Thai, or Caribbean cuisines
· Spices and spice blends, especially curry powder mixtures containing turmeric
· Turmeric-based pickles, which are common in some cultures
· Turmeric-containing supplements, available in various forms such as drinks, capsules, or fortified nutritional products
It is important to note that while these sources provide some curcumin, the amounts are generally low compared to concentrated supplements. The bioavailability of curcumin from dietary sources is also limited, which may affect its potential health benefits when consumed in food form.102
Supplemental Sources
Turmeric supplements are available in various forms, including capsules, tablets, powders, and liquid extracts. Some formulations may include additional compounds like piperine to enhance bioavailability. Generally, standardized extracts may be more effective than raw turmeric powder due to their higher curcumin content.103 Some supplements provide up to 1,000 mg of curcumin per serving and may use specific forms of curcumin, such as the C3 complex, which is well-studied.104 Many formulations also include complementary ingredients to enhance effectiveness. For example, some supplements combine turmeric with ginger for added anti-inflammatory benefits, while others include bromelain, an enzyme that can increase the absorption and anti-inflammatory effects of curcumin.
Side Effects
Common side effects of high doses may include gastrointestinal issues such as nausea, diarrhea, and stomach upset.105 Curcumin may interact with blood thinners and diabetes medications. Pregnant women should consult a healthcare provider before using turmeric supplements.106
Bottom Line
While turmeric and curcumin show promising potential in various health applications, particularly for their anti-inflammatory and antioxidant properties, more high-quality clinical trials are needed to establish their efficacy conclusively. Current evidence supports their use as a complementary approach in managing certain inflammatory conditions, but they should not replace conventional medical treatments without professional guidance.
Specific Nutrient: Inositol
What Is It
Inositol is a naturally occurring sugar alcohol found in both plants and animals.107 Its scientific name is cyclohexane-1,2,3,4,5,6-hexol, with the most common form being myo-inositol.108 Historically, inositol was considered part of the vitamin B complex (vitamin B8), but it is now recognized as a pseudovitamin since the human body can synthesize it.109 Inositol plays crucial roles in cellular signaling, osmoregulation, and as a structural component of cell membranes.110
How It Works
Inositol plays a crucial role as a second messenger in cellular signaling pathways, particularly as a component of the phosphatidylinositol (PI) signaling system.111 When certain receptors are activated, phospholipase C hydrolyzes phosphatidylinositol 4,5-bisphosphate (PIP2) into two second messengers: diacylglycerol (DAG) and inositol 1,4,5-trisphosphate (IP3).112 IP3 acts as a key second messenger by diffusing through the cytoplasm and binding to IP3 receptors on the endoplasmic reticulum, triggering the release of calcium ions (Ca2+) from intracellular stores, which leads to various cellular responses.113
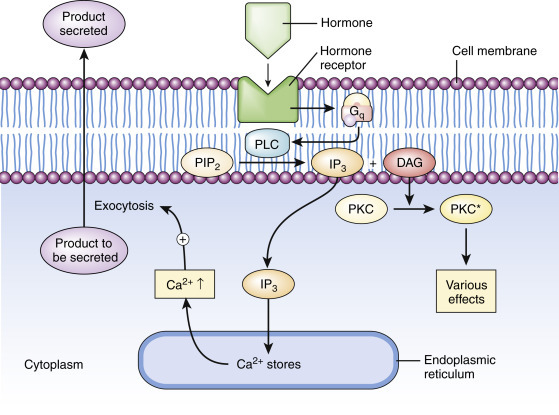
Figure 2 IP3 and DAG as secondary messengers, source: DOI:10.1016/B978-0-12-394815-1.00003-3
Inositol and its derivatives, particularly myo-inositol and D-chiro-inositol, are integral to insulin signaling and glucose metabolism.114 They exhibit insulin-mimetic activity through inositol phosphoglycans (IPGs), which act as second messengers downstream of insulin receptors, mimicking some of insulin’s actions. Additionally, myo-inositol improves muscle glucose uptake and inhibits duodenal glucose absorption, helping to regulate blood glucose levels in PCOS cases.115 It also promotes glycogen storage in muscle tissue, further contributing to glucose metabolism. Both myo-inositol and D-chiro-inositol are involved in the intracellular transmission of insulin’s metabolic signals, enhancing insulin sensitivity.
In the central nervous system, inositol influences neurotransmitter signaling, which may explain its effects on mood regulation in individuals with PCOS.116 It can affect the activity of key neurotransmitters such as serotonin and dopamine, which are involved in mood, anxiety, and cognitive function.117 Furthermore, inositol derivatives like IP3 act as second messengers in various neurotransmitter pathways, influencing neuronal signaling. This modulation of neurotransmitter activity suggests that inositol may have therapeutic potential in managing mood disorders.118
Inositol’s effects on hormone balance are particularly relevant in conditions like polycystic ovary syndrome. Myo-inositol supplementation has been shown to reduce serum androgen levels in women with PCOS.119 Additionally, inositol phosphoglycans serve as secondary messengers for follicle-stimulating hormone (FSH) and thyroid-stimulating hormone (TSH), influencing reproductive and thyroid function. These diverse mechanisms highlight inositol’s wide-ranging effects on cellular function, metabolism, and neurological processes, explaining its potential therapeutic applications in various conditions, from metabolic disorders to mental health issues.
What the Research Is Telling Us
Extensive research has been conducted on inositol, particularly regarding its effects on PCOS and fertility. A prospective clinical trial evaluated the effectiveness of myo-inositol (MI) in treating polycystic ovary syndrome over an 18-month period.120 The researchers enrolled 90 women diagnosed with PCOS, administering 1 gram of myo-inositol twice daily for six months. Before beginning treatment, the team recorded baseline clinical, hormonal, and metabolic parameters for each participant. These same parameters were reassessed at the conclusion of the six-month treatment period to measure the effects of MI supplementation. The study’s results were promising, showing significant improvements in several key areas. Notably, 68% of participants experienced a return to regular menstrual cycles. Hormonal improvements were evident, with luteinizing hormone (LH) levels decreasing from 10.31 ± 7.92 to 7.42 ± 6.25 (p = 0.002), and the LH/FSH ratio dropping from 2.34 ± 0.34 to 1.91 ± 0.32 (p < 0.001). Metabolic benefits were also observed, with significant reductions in fasting serum insulin levels and HOMA-IR scores, indicating enhanced insulin sensitivity. These findings led the researchers to conclude that myo-inositol is an effective treatment for improving the hormonal and metabolic profiles of women with PCOS, offering good safety and compliance. The study supports the use of myo-inositol as a viable option for managing PCOS symptoms, particularly menstrual irregularities and insulin resistance.
One meta-analysis examined the impact of myo-inositol, either alone or in combination with d-chiro-inositol (DCI), on endocrine and metabolic abnormalities in women with PCOS.121 Researchers systematically reviewed several databases (MEDLINE, EMBASE, PubMed, and ResearchGate) for relevant randomized controlled trials (RCTs). The meta-analysis included nine RCTs, with a total of 247 women with PCOS who received MI (alone or with DCI) and 249 control participants who received either folic acid (FA) or other treatments, such as DCI or oral contraceptive pills (OCPs). The studies, conducted in Italy, Georgia, and Turkey, were published between 2007 and 2016. The daily dose of MI ranged from 1.1g to 4g, and treatment duration varied from 12 to 24 weeks.
The primary outcome of interest was fasting insulin levels, while secondary outcomes included the homeostasis model assessment (HOMA) index, testosterone levels, androstenedione levels, and sex hormone-binding globulin (SHBG) plasma levels. The results showed that MI supplementation significantly decreased fasting insulin levels and the HOMA index in women with PCOS. A slight trend toward reduced testosterone levels was observed, though it was not statistically significant. However, subgroup analysis indicated a significant increase in serum SHBG levels in studies where MI was administered for at least 24 weeks. The authors concluded that MI is beneficial in improving the metabolic profile of women with PCOS and in reducing hyperandrogenism.
A study by Victoria Fitz et al., published in The Journal of Clinical Endocrinology & Metabolism, was a systematic review and meta-analysis that evaluated the efficacy of inositol (myo-inositol and D-chiro-inositol) for managing PCOS. The findings informed the 2023 update of the International Evidence-Based PCOS Guidelines.122 The study aimed to determine the effectiveness of inositol, alone or combined with other therapies, on various outcomes in women with PCOS, including metabolic, hormonal, reproductive, psychological, and anthropometric measures. The analysis included 30 trials with 2,230 participants (1,093 intervention, 1,137 control), 19 of which were pooled for meta-analysis. Outcomes assessed included hormonal levels, metabolic parameters (e.g., insulin resistance), lipid profiles, psychological measures, anthropometric data (e.g., BMI), reproductive outcomes (e.g., ovulation), and adverse effects.
The study found that both myo-inositol and D-chiro-inositol provided some benefits for metabolic measures, such as improving insulin sensitivity. D-chiro-inositol showed potential benefits for ovulation. Compared to metformin, a commonly used medication for PCOS, metformin was more effective in improving waist-to-hip ratio and reducing hirsutism. However, myo-inositol caused fewer gastrointestinal side effects than metformin. No significant differences were observed between inositol and metformin for reproductive outcomes. Inositol was generally associated with mild, self-limited gastrointestinal side effects. The study concluded that while inositol may offer some benefits for metabolic parameters and ovulation in women with PCOS, its overall clinical efficacy remains uncertain.
Contradictory findings in regards to reproductive outcomes, particularly menstrual regulation, were presented in a study by Dorina Greff et al., published in Reproductive Biology and Endocrinology.123 This systematic review and meta-analysis evaluated the efficacy and safety of inositol as a treatment for PCOS. The meta-analysis included 26 randomized controlled trials (RCTs) with a total of 1,691 participants—806 receiving inositol, 311 receiving placebo, and 509 treated with metformin. Results demonstrated that inositol significantly improved menstrual cycle regularity compared to placebo, with a relative risk of 1.79 indicating a 79% increase of reported improvement of cycle regularity. Inositol also reduced body mass index (BMI), particularly when myo-inositol was used, and decreased levels of total testosterone, free testosterone, androstenedione, fasting glucose, and insulin resistance markers. Additionally, inositol increased sex hormone-binding globulin (SHBG) levels compared to placebo. The study concluded that inositol is non-inferior to metformin for most outcomes but causes fewer gastrointestinal side effects. Overall, it was deemed an effective and safe treatment option for women with PCOS, showing significant benefits for metabolic and hormonal parameters while improving menstrual cycle regularity.
Regarding inositol’s use in fertility treatments for women with PCOS, a comprehensive systematic review was conducted to assess its effectiveness and safety for subfertile women with PCOS.124 This review examined evidence from randomized controlled trials (RCTs) comparing inositol (myo-inositol, D-chiro-inositol, or a combination) to placebo, no treatment, metformin, or other interventions in subfertile women with PCOS. Primary outcomes of interest included live birth rate, pregnancy rate, and adverse events, with secondary outcomes covering ovulation, menstrual regularity, hormonal and metabolic parameters, and multiple pregnancy rates. The review found limited evidence on live birth rates, which was insufficient to draw firm conclusions. However, it suggested that inositol may be associated with increased pregnancy rates compared to placebo or no treatment, though the quality of the evidence was low. Inositol appeared to improve ovulation rates and was generally well-tolerated with a low risk of adverse effects. Evidence comparing inositol to metformin was limited and inconclusive. The authors concluded that while there is some low-quality evidence suggesting inositol may improve pregnancy and ovulation rates in subfertile women with PCOS, the evidence is not strong enough for definitive recommendations. They emphasized the need for further high-quality RCTs with larger sample sizes to confirm these findings, compare inositol to other common fertility treatments for women with PCOS, and assess live birth rates in future research. Research is also needed to assess the safety of inositol use during pregnancy.
Dosage Recommendation Based on Research
Dosage recommendations for inositol vary depending on the condition being treated. For PCOS, doses of 2-4 grams per day have shown efficacy.125
Table 5 Dosage Recommendation for Inositol
| What’s the Daily Recommended Intake? | How much do I take? | Don’t take more than… | When will I see results? |
|---|---|---|---|
| NA | 2-4g per day for PCOS | 12-18 g per day | 8-12 weeks minimum |
Facts and Misconceptions
Inositol, often referred to as a pseudovitamin, is not a true vitamin because the human body can synthesize it, primarily in the kidneys, from glucose. Unlike essential vitamins that must be obtained through diet, inositol is naturally produced and does not lead to deficiency-related diseases if absent in dietary intake. While it was historically classified as part of the vitamin B complex (vitamin B8), this designation is no longer accurate.
A common misconception is that inositol is only beneficial for women with polycystic ovary syndrome. While it has shown significant benefits for managing PCOS symptoms, such as improving insulin sensitivity, reducing androgen levels, and enhancing ovarian function, its applications extend far beyond this condition. Research has demonstrated its potential in addressing metabolic disorders like type 2 diabetes and metabolic syndrome by improving blood sugar control and increasing insulin sensitivity.
Additionally, there is a misconception that all forms of inositol are equally effective. Myo-inositol and D-chiro-inositol are the two primary biologically active forms, each with distinct roles. Myo-inositol is particularly effective for improving ovarian function and metabolic parameters in PCOS, while D-chiro-inositol plays a more prominent role in enhancing insulin sensitivity. The balance between these two forms is critical for optimal therapeutic outcomes.126
Food Sources
Fruits
o Orange: 307 mg per 1 medium fruit (100g)
o Grapefruit: 199 mg per 1/2 fruit (100g)
o Cantaloupe: 355 mg per 1 cup serving
o Kiwi: 136 mg per 1/2 cup (100g)
o Watermelon: 62 mg per 1 cup (100g)
· Beans and Legumes
o Great Northern Beans: 440 mg per 1/2 cup serving (canned)
o Green Beans: 105 mg per 1/2 cup (100g)
o Kidney Beans: 249 mg per 1/2 cup (canned)
o Chickpeas, lentils, and soybeans: High in inositol, specific amounts vary
· Grains
o Stone-ground whole wheat bread: 287.5 mg per 1 medium slice
o Bran flakes: 54.8 mg per 1/2 cup (20g)
o Cooked oatmeal (instant): 34 mg per 1/2 cup (100g)
· Nuts
o Almonds: 41.7 mg per 8 pieces (15g)
o Peanuts: 20 mg per 12 pieces (15g)
o Cashews: 12.2 mg per 5 pieces (15g)
· Beef and Liver
o Beef liver (raw): Rich in inositol, specific amount not provided
o Chicken liver: 39 mg per serving, specific serving size not provided
· Dairy
o Plain yogurt: 14.4 mg per 1 cup (240g)
o Chocolate milk (low fat): 45.6 mg per 1 cup (240g)
o Cheddar cheese: 2.7 mg per 1 oz (30g)
Supplemental Sources
Inositol supplements are available in powder, capsule, and tablet forms. The powder form is often preferred for higher doses due to ease of administration. There is no clear evidence suggesting that supplements are more effective than dietary sources, but supplements allow for precise dosing and higher concentrations than typically obtained through diet alone.
Side Effects
Common side effects of high-dose inositol supplementation may include mild gastrointestinal disturbances such as nausea, gas, and diarrhea. No significant drug interactions have been reported, but as with any supplement, it is advisable to consult a healthcare provider before use, especially for individuals on medications or with pre-existing conditions.
Bottom Line
Inositol shows promise as a therapeutic agent, particularly for PCOS and certain metabolic disorders. While research is encouraging, especially for reproductive health, the evidence for its efficacy in other conditions is still emerging. The strength of recommendation for inositol use in PCOS management is moderate, based on consistent findings from multiple studies. However, for other applications, the strength of recommendation is low to moderate, pending further high-quality research. As with any supplement, individuals should consult healthcare providers before incorporating inositol into their health regimen.
Specific Nutrient: Berberine
What Is It
Berberine is an isoquinoline alkaloid compound found in various plants, including Berberis species, Hydrastis canadensis (goldenseal), and Coptis chinensis.127 Its scientific name is 10-Dimethoxy-7,8,13,13a-tetradehydro-2′H-dioxolo[4′,5′:2,3]berbin-7-ium. Berberine has been used for thousands of years in traditional Chinese and Ayurvedic medicine to treat various conditions, including infections, diarrhea, and inflammatory disorders.128
How It Works
Berberine’s mechanism of action involves multiple pathways and physiological processes seen in PCOS, primarily affecting glucose and lipid metabolism. It improves glucose metabolism through several key mechanisms. For instance, berberine activates AMP-activated protein kinase (AMPK), a crucial regulator of cellular energy homeostasis.129 This activation leads to increased glucose uptake in cells, enhanced insulin sensitivity, and improved translocation of glucose transporter 4 (GLUT4) to the plasma membrane.130 Additionally, berberine stimulates glycolysis by increasing glucokinase activity and inhibiting oxygen-dependent glucose oxidation in mitochondria, thereby enhancing anaerobic respiration, which requires more glucose for ATP production.131
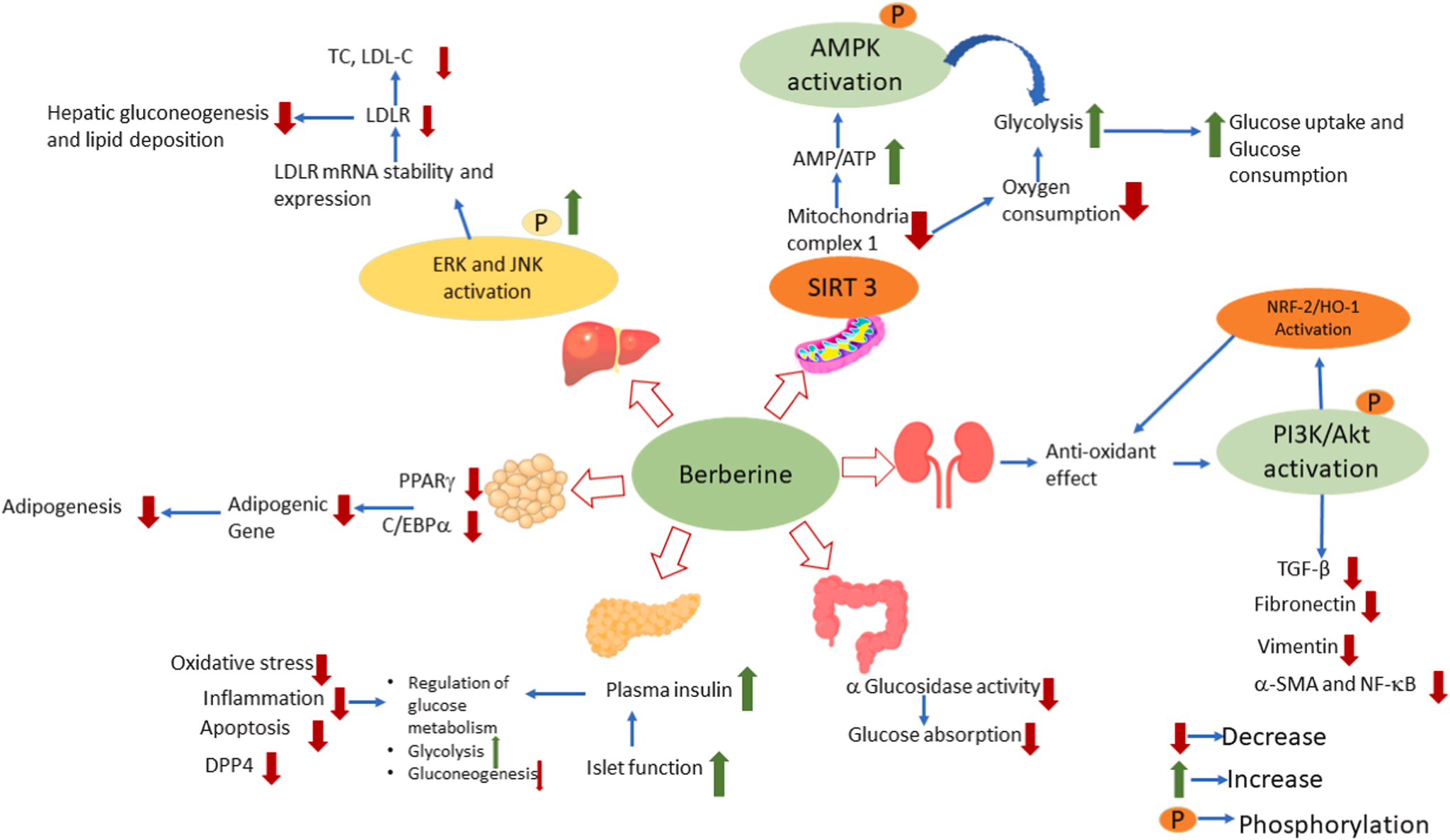
Figure 3 Molecular targets and mechanism of action of berberine in T2DM and associated complications, source: DOI:10.1016/j.heliyon.2023.e21233
Berberine helps regulate blood sugar by both increasing insulin secretion and improving insulin sensitivity, particularly in people with insulin resistance, such as those with PCOS. It increases Akt phosphorylation, activating the Akt signaling pathway, which is vital for insulin sensitivity and glucose metabolism.132 Furthermore, berberine suppresses glucose production in the liver by inhibiting gluconeogenesis, a process that generates glucose from non-carbohydrate sources. In terms of lipid metabolism, berberine activates AMPK in liver cells, inhibiting cholesterol and triglyceride synthesis. It enhances expression of AMPK-dependent adipose tissue triglyceride lipase, promoting long-term weight loss by increasing lipolysis.133 Berberine also reduces fat accumulation by inhibiting adipogenesis in the liver.134
At the cellular level, berberine affects energy regulation by reducing oxygen consumption in cells and inhibiting NAD-linked respiration in mitochondria. This shift in energy production leads to increased glucose utilization through glycolysis.135 Berberine may also activate the AMPK-p38 pathway, although this is less well understood, contributing to increased glucose uptake in muscle cells.136
Berberine’s interaction with gut microbiota plays a significant role in its metabolism and absorption. Gut bacteria convert berberine to dihydroberberine (dhBBR), which has higher intestinal absorption.137 Other metabolites produced by gut bacteria, such as berberrubine, demethyleneberberine, jatrorrhizine, and thalifendine, also contribute to berberine’s effects.138 These complex interactions and pathways contribute to berberine’s overall effects on glucose and lipid metabolism, making it a potential therapeutic agent for metabolic disorders such as PCOS. However, the relative importance of each pathway and the interplay between them are still subjects of ongoing research.
What the Research Is Telling Us
Recent studies have provided valuable insights into the potential benefits of berberine for managing polycystic ovary syndrome. A randomized controlled trial comparing berberine to metformin in women with PCOS found that both treatments had similar effects on androgen levels, reducing testosterone and lowering the free androgen index.139 The study examined the comparative effects of berberine, metformin, and myoinositol in 136 women aged 15-40 diagnosed with PCOS, excluding those with endocrine disorders, diabetes, or medications that could alter insulin function. Participants were randomly assigned to one of three treatment groups: berberine hydrochloride (500 mg twice daily), metformin hydrochloride (500 mg twice daily), or myoinositol (1000 mg twice daily). The study lasted for three months without specific lifestyle recommendations. Researchers assessed clinical, metabolic, hormonal, and lipid profile parameters before and after treatment. Of the 136 participants, 129 completed the study (43 per group). The results showed statistically significant improvements across a range of parameters, including weight, BMI, waist circumference, waist-to-hip ratio, fasting blood sugar, fasting insulin, glucose/insulin ratio, total testosterone, sex hormone-binding globulin, free androgen index, and lipid profile markers. Notably, berberine showed greater improvements in clinical, hormonal, and lipid parameters compared to metformin and myoinositol. These findings suggest that berberine could be a viable alternative to metformin for managing PCOS symptoms, particularly for those who may not tolerate metformin well.
A narrative review of five studies involving 1078 women also highlighted berberine’s potential in treating insulin resistance and obesity, particularly visceral adipose tissue (VAT).140 Some studies indicated that berberine helps redistribute adipose tissue, reducing VAT without causing overall weight loss, and improving insulin sensitivity, similar to metformin. One study showed berberine also improved lipid profiles. Additionally, some research suggested that berberine could improve insulin resistance in theca cells (endocrine cells within the ovary that play a crucial role in ovarian function, particularly during folliculogenesis), potentially enhancing ovulation rates, fertility, and live birth rates.
A multicenter, randomized, double-blinded, placebo-controlled trial with 644 infertile women with PCOS found that berberine combined with letrozole (a hormone-based chemotherapy that lowers the levels of estrogen in the body) did not significantly affect ovulation or live birth rates compared to letrozole alone.141 This mixed effectiveness underscores the need for further research to understand berberine’s role in reproductive health.
In addition to its metabolic and hormonal benefits, berberine has been shown to improve dermatological symptoms associated with PCOS. The study by Rondanelli et al., which was the first to assess acne severity using standardized tools in PCOS patients treated with berberine, reported improvements in acne, with patients moving from “Moderate” to “Mild” on the Global Acne Grading System.142 This suggests that berberine may help manage acne, a common symptom of PCOS.
Comparative studies have also explored berberine’s efficacy relative to other treatments. A meta-analysis and systematic review found no significant difference between berberine and metformin in improving insulin resistance, glycolipid metabolism, or reproductive endocrine conditions.143 However, the combination of cyproterone acetate (progestin and antiandrogen hormonal therapy) and berberine showed superior results in improving some reproductive endocrine indices compared to cyproterone acetate alone. Furthermore, a comparison study of berberine, myoinositol, and metformin in PCOS patients suggested that berberine may offer greater potential to reduce cardiovascular disease risk in PCOS patients, due to its effects on body composition, lipid profile, and hormone status.144
Despite these promising findings, researchers stress the need for more rigorously designed, randomized, double-blind, placebo-controlled trials to confirm berberine’s effects and safety in PCOS management.145 While multiple studies report few side effects with berberine, indicating its safety for premenopausal women (including those seeking pregnancy), further long-term studies are necessary to establish its safety profile for extended use.146
Dosage Recommendation Based on Research
Based on clinical studies, the typical dosage of berberine ranges from 900 to 1500 mg per day, usually divided into 2-3 doses.147 However, optimal dosage may vary depending on the specific condition being treated and individual factors.
Table 6 Dosage Recommendation for Berberine
| What’s the Daily Recommended Intake? | How much do I take? | Don’t take more than… | When will I see results? |
|---|---|---|---|
| NA | 900-1500mg per day | 2000mg per day | 4-12 weeks |
Facts and Misconceptions
One common misconception is that berberine is a cure-all for metabolic disorders. While it shows promise, it should be considered as part of a comprehensive treatment plan, not a standalone solution. Additionally, berberine’s low bioavailability is often misunderstood; its effects are largely mediated through gut microbiota modulation rather than direct absorption into the bloodstream.148
Food Sources
Berberine is not typically found in common foods. It is primarily obtained from medicinal plants such as:
Berberis vulgaris (Barberry): 28 grams (1 ounce) of dried barberries.
Hydrastis canadensis (Goldenseal): 500–1000 mg per day, often divided into two doses.
Coptis chinensis (Chinese Goldthread): 500 mg per day.
Mahonia aquifolium (Oregon Grape): 300–500 mg per day.
Supplemental Sources
Berberine is most commonly available as a dietary supplement in capsule or tablet form. There is limited evidence suggesting significant differences in efficacy between these forms.149 Supplements are generally considered more concentrated and consistent in dosage compared to raw plant sources.
Side Effects
Common side effects of berberine include gastrointestinal discomfort, constipation, and diarrhea.150 Berberine may interact with certain medications, particularly those metabolized by cytochrome P450 enzymes.151 It should be used with caution in pregnant or breastfeeding women due to potential risks.152
Bottom Line
Berberine shows promising potential for managing various metabolic disorders, particularly type 2 diabetes and dyslipidemia. However, while many studies demonstrate positive effects, the overall evidence is still evolving. The strength of recommendation for berberine use is moderate, based on the current body of evidence. Healthcare professionals should consider berberine as a potential adjunct therapy for certain metabolic conditions, but not as a replacement for established treatments. Patients should be advised to use berberine under medical supervision, especially if they have pre-existing health conditions or are taking other medications.
Dietary Strategy
Dietary interventions have emerged as a cornerstone in the management of polycystic ovarian syndrome, with various research diets demonstrating significant improvements in metabolic and hormonal outcomes. Several dietary methods, including low glycemic index (GI) diets, Mediterranean diets, ketogenic diets, high-protein, low-carbohydrate diets, and those emphasizing the inclusion of lean protein, have been explored for their efficacy in managing PCOS. These interventions target key mechanisms underlying PCOS, such as insulin resistance, chronic inflammation, and hormonal imbalances, offering non-pharmacological strategies to alleviate symptoms and improve quality of life.
Low Glycemic Index Diet
One dietary approach that has been extensively studied in PCOS management is the low glycemic index (GI) diet. The glycemic index (numerical scale from 0 to 100) is a ranking system for carbohydrates based on their immediate effect on blood glucose levels.153 Foods with a low GI value (55 or less) are often whole or minimally processed, high in fiber, and are digested and absorbed more slowly, resulting in a gradual rise in blood sugar and insulin levels, while high-GI foods (70 or more) cause rapid spikes.154 The concept of GI was originally developed to help individuals with diabetes manage their blood sugar levels.155 However, it has since gained broader application in nutrition and health management.
The gradual glucose release reduces stress on the body’s glucose regulation mechanisms, benefiting individuals with diabetes or conditions like PCOS, and promotes satiety, aiding in weight management by reducing calorie intake.156 Additionally, low GI foods provide steady energy throughout the day, preventing the rapid spikes and crashes associated with high GI foods, while potentially encouraging fat oxidation due to lower insulin levels.157 By reducing glycemic variability and influencing hormones like GLP-1, which regulate appetite and glucose homeostasis, low GI foods offer long-term benefits for metabolic health, weight management, and overall well-being. Incorporating them into a diet can be an effective strategy for maintaining stable blood sugar and supporting overall health.
Examples of low-GI foods include most non-starchy vegetables, such as broccoli, spinach, and carrots; whole grains like quinoa, brown rice, and oats; legumes such as lentils, chickpeas, and beans; nuts and seeds; and certain fruits like apples, berries, and pears. These foods are digested slowly, preventing drastic glucose surges.
Table 7 Glycemic Index Rating Per Food Item
| Low GI (55 or less) | Medium GI (56-69) | High GI (70-100) |
|---|---|---|
| Soya beans (16) | Potato chips (56) | Unleavened wheat bread (70) |
| Kidney beans (24) | Muesli (57) | White rice, boiled (73) |
| Barley (28) | Pineapple (59) | Whole wheat bread (74) |
| Chickpeas (28) | Soda, non-diet (59) | White bread (wheat) (75) |
| Lentils (32) | Wheat roti (62) | Watermelon (76) |
| Soy milk (34) | French fries (63) | Potato, boiled (78) |
| Apple (36) | Sweet potato, boiled (63) | Rice porridge (78) |
| Skim milk (37) | Pumpkin, boiled (64) | Instant oatmeal (79) |
| Whole milk (39) | Couscous (65) | Cornflakes (81) |
| Carrots, boiled (39) | Popcorn (65) | Rice milk (86) |
| Chocolate (40) | Millet porridge (67) | Potatoes, instant mashed (87) |
| Apple juice (41) | Brown rice, boiled (68) | Rice crackers (87) |
| Yogurt, fruit (41) | Wheat flake biscuits cereal (69) | Baguette (95) |
| Dates (42) | Raisins (64) | Parsnips (97) |
| Orange (43) | Instant noodles (65) | Glucose (100) |
| Peaches, canned (43) | Basmati rice (58) | Jasmine rice (89) |
| Spaghetti, white (49) | Honey (61) | Puffed wheat cereal (80) |
| Spaghetti, whole grain (48) | Rye bread (65) | Pretzels (83) |
| Vegetable soup (48) | Cantaloupe (65) | Jelly beans (80) |
| Orange juice (50) | Quick oats (66) | Bagel (72) |
| Banana (51) | Croissant (67) | Doughnuts (75) |
| Ice cream (51) | Pasta, white (58) | Waffles (76) |
| Mango (51) | Sushi rice (67) | Tapioca (81) |
| Chapatti (52) | Pita bread (57) | Cheerios (74) |
| Sweet corn (52) | Bulgur (58) | Russet potato (85) |
| Rice noodles (53) | Hamburger bun (61) | Gatorade (78) |
| Specialty grain bread (53) | Beets (64) | Table sugar (sucrose) (65) |
| Taro, boiled (53) | Corn tortilla (60) | White potato (82) |
| Plantain (55) | Macaroni and cheese (64) | Maltose (105) |
| Rolled oats (55) | Taco shells (68) | Potato, baked (85) |
| Udon noodles (55) | Canned peaches (67) | French baguette (95) |
Research indicates that following a low-GI diet can lead to significant improvements in PCOS-related outcomes. For instance, one study where participants followed a low-GI diet for 12 months showed improvements in insulin sensitivity, menstrual regularity, and emotional well-being compared to those following a conventional healthy diet.158 Similarly, research has found that a low-GI diet led to greater reductions in fasting insulin levels and free androgen index compared to a standard healthy diet, despite both groups achieving similar weight loss.159 By focusing on foods that promote stable blood glucose levels and reduce insulin resistance, low-GI diets can play a critical role in mitigating hormonal imbalances and improving metabolic health in women with PCOS. This approach, therefore, represents a sustainable dietary strategy for long-term management of the condition.
High Protein, Low Carbohydrate Diet
High-protein, low-carbohydrate diets have also been explored for their potential benefits in PCOS management. These diets involve increasing the proportion of protein in the diet while significantly reducing carbohydrate intake. The goal is to shift the body’s primary fuel source from carbohydrates to fat, promoting weight loss and improving metabolic markers.160 A high-protein diet typically derives more than 40% of its energy from protein, with the remaining energy coming from fats and a small percentage from carbohydrates, often less than 30% of total calories.
Common food choices in high-protein, low-carbohydrate diets include lean meats such as chicken, turkey, fish, and lean cuts of beef; eggs; low-fat dairy products like Greek yogurt and cottage cheese; and plant-based protein sources such as tofu, tempeh, and legumes (in moderation due to their carbohydrate content). Non-starchy vegetables like leafy greens, broccoli, and cauliflower are also included to provide essential micronutrients and fiber. Foods that are typically restricted include grains (bread, pasta, rice), sugary beverages, sweets, starchy vegetables (potatoes, corn), and most fruits due to their high carbohydrate content.
Sørensen et al. investigated the effects of a high-protein diet (with more than 40% of energy derived from protein) compared to a standard protein diet in women with PCOS over six months.161 The high-protein group experienced greater weight loss, improved insulin sensitivity, and reduced testosterone levels. Protein-rich diets are thought to be effective due to their satiating properties and higher thermic effect, which can aid in weight management, a key factor in improving PCOS symptoms. The enhanced satiety from protein can help reduce overall calorie intake, while the higher thermic effect means the body burns more calories during digestion, absorption, and metabolism of protein compared to carbohydrates or fats. Further research supported these findings in their, concluding that high-protein diets may be more effective than standard diets for achieving metabolic improvements in women with PCOS.162
Mediterranean Diet
The Mediterranean diet has gained attention as another promising dietary intervention for PCOS due to its anti-inflammatory and antioxidant properties. This dietary pattern is rooted in the traditional eating habits of countries bordering the Mediterranean Sea, emphasizing the consumption of whole, unprocessed foods. Key components include abundant fruits and vegetables (such as tomatoes, leafy greens, peppers, and citrus fruits), whole grains (like brown rice, quinoa, and whole wheat bread), legumes (such as lentils, chickpeas, and beans), nuts and seeds (like almonds, walnuts, and flaxseeds), olive oil as the primary source of fat, and moderate amounts of fish and dairy (primarily yogurt and cheese). Red meat and processed foods are limited, and poultry is consumed in moderation.
The benefits of the Mediterranean diet in PCOS management stem from several factors. Barrea et al. found that women with PCOS who adhered closely to the Mediterranean diet had lower body mass index, improved insulin sensitivity, and better hormonal profiles compared to those with lower adherence.163 The high fiber content of this diet likely contributes to its benefits by improving satiety, regulating blood sugar levels, and promoting gut health. Additionally, the Mediterranean diet’s rich supply of antioxidants, derived from its emphasis on colorful fruits and vegetables and healthy fats like olive oil, may help reduce the chronic low-grade inflammation often observed in women with PCOS. The combination of these factors makes the Mediterranean diet a potentially sustainable and effective dietary strategy for improving metabolic and hormonal health in women with PCOS.
Ketogenic Diet
The ketogenic diet has also been explored for its potential role in managing PCOS symptoms. This very low-carbohydrate, high-fat diet induces ketosis, a metabolic state where the body burns fat for energy instead of carbohydrates.164 The typical macronutrient breakdown in a ketogenic diet is around 70-80% fat, 20-25% protein, and 5-10% carbohydrates. By drastically reducing carbohydrate intake, the body switches from using glucose as its primary fuel source to using ketones, which are produced from the breakdown of fats in the liver. This metabolic shift has shown promise in addressing hyperinsulinemia and weight loss, two common challenges for women with PCOS.165
Foods commonly included in a ketogenic diet are high in healthy fats, such as avocados, nuts and seeds (like almonds, walnuts, and flaxseeds), olive oil, coconut oil, and fatty fish like salmon and mackerel. Protein sources include meat, poultry, eggs, and seafood, all of which should ideally be lean to moderate in fat content. Non-starchy vegetables like leafy greens (spinach, kale), broccoli, cauliflower, and bell peppers are also included in limited quantities to provide essential micronutrients. Conversely, foods high in carbohydrates, such as grains (rice, bread, pasta), starchy vegetables (potatoes, corn), fruits (especially high-sugar varieties like bananas and grapes), legumes, and sugary snacks and beverages are strictly limited or avoided altogether to maintain ketosis. A pilot study was conducted on women with PCOS following a ketogenic diet for 24 weeks, and participants experienced significant reductions in body weight, fasting insulin levels, and free testosterone levels.166 However, while these findings are promising, adherence to the ketogenic diet can be challenging due to its restrictive nature. Additionally, concerns about long-term safety remain unresolved, emphasizing the need for further research to evaluate its efficacy over extended periods.
The ketogenic diet has also been explored for its potential role in managing PCOS symptoms. This very low-carbohydrate, high-fat diet induces ketosis, a state where the body burns fat for energy instead of carbohydrates, and has shown promise in addressing hyperinsulinemia and weight loss. A pilot study was conducted on women with PCOS following a ketogenic diet for 24 weeks.167 Participants experienced significant reductions in body weight, fasting insulin levels, and free testosterone levels. However, while these findings are promising, adherence to the ketogenic diet can be challenging due to its restrictive nature. Additionally, concerns about long-term safety remain unresolved, emphasizing the need for further research to evaluate its efficacy over extended periods.
Lean Protein
Incorporating lean protein into PCOS patients’ diets has shown promising results in recent research. A clinical trial involving 57 women with PCOS demonstrated that a high-protein diet (more than 40% of energy from protein) led to greater weight loss, body fat reduction, and decreased waist circumference compared to a standard-protein diet over a 6-month period.168 Importantly, the high-protein diet resulted in significant reductions in glucose levels, which persisted even after adjusting for weight changes. This suggests that replacing carbohydrates with protein in ad libitum diets can improve both weight loss and glucose metabolism in women with PCOS, independent of weight loss alone. Furthermore, a systematic review and meta-analysis of eight clinical trials involving 300 women with PCOS found that high-protein diets significantly reduced fasting insulin levels and lowered the homeostasis model assessment for insulin resistance (HOMA-IR) compared to balanced diets.169 While no significant differences were observed in weight loss, abdominal fat, lipid profiles, or reproductive hormones, the improvement in insulin resistance suggests that high-protein diets could be a viable dietary management option for PCOS. Incorporating lean protein into the diet is essential for managing polycystic ovarian syndrome, as it supports weight management, improves insulin sensitivity, and helps regulate glucose levels.
Lean protein refers to sources that are low in fat and calories while providing essential amino acids necessary for various bodily functions. Examples of animal-based lean protein include chicken breast, turkey, lean beef tenderloin, fish like tilapia or cod, pork tenderloin, and eggs. These options are rich in protein yet low in saturated fat and cholesterol, making them ideal for promoting metabolic health. Plant-based lean protein sources such as lentils, chickpeas, tofu, tempeh, edamame, quinoa, hemp seeds, and beans are excellent alternatives. These options not only provide protein but also offer additional nutrients like fiber and antioxidants that can help reduce inflammation often associated with PCOS.
Supplemental sources of lean protein can also be beneficial for individuals with busy lifestyles or specific dietary needs. Whey protein is a popular choice due to its rapid absorption and complete amino acid profile, making it ideal for post-workout recovery. Other options include casein protein for slow digestion, soy protein for vegetarians and vegans, pea protein for those with allergies, and hemp protein for added omega-3 fatty acids. While supplements can help meet daily protein requirements, they should complement whole food sources rather than replace them entirely to ensure a balanced intake of nutrients. By incorporating a variety of lean protein sources into the diet—both animal- and plant-based—women with PCOS can better manage their symptoms while improving overall metabolic health and hormone regulation.170
The collective findings from these studies underscore the importance of dietary interventions tailored to address the specific metabolic and hormonal challenges associated with PCOS. Low-GI diets are particularly effective for improving glycemic control and reducing androgen excess; high-protein diets aid in weight management and metabolic regulation; the Mediterranean diet offers anti-inflammatory benefits while promoting overall health; ketogenic diets show promise but require careful monitoring; and high-fiber diets contribute significantly to improving insulin sensitivity and gut health.
While these dietary approaches demonstrate significant potential for managing PCOS symptoms, it is important to acknowledge that individual responses can vary based on factors such as body composition, metabolic status, lifestyle habits, and personal preferences. A personalized approach that combines dietary modifications with other lifestyle interventions, such as regular physical activity and stress management, may yield the most effective outcomes for women with PCOS.
Future research should focus on long-term studies to assess the sustainability of these dietary interventions while exploring potential synergistic effects when combining different approaches. For instance, integrating elements of a low-GI or high-fiber diet into a Mediterranean dietary framework could provide comprehensive benefits by targeting multiple aspects of PCOS pathophysiology simultaneously. Additionally, investigating how dietary changes influence gut microbiota composition could offer new insights into the mechanisms underlying these effects and open up novel therapeutic strategies for managing this complex condition effectively.
Weight Management and the Role of Exercise in PCOS
Effective weight management and regular physical activity are fundamental components in the management of polycystic ovary syndrome. Together, these lifestyle interventions address the metabolic, hormonal, and psychological challenges associated with the condition, offering a comprehensive approach to symptom relief and overall health improvement.
Even modest weight loss, defined as a reduction of 5-10% of total body weight, can yield significant benefits for women with PCOS.171 Insulin resistance plays a critical role in driving hyperinsulinemia and exacerbating hormonal imbalances, such as elevated androgen levels. Studies have shown that losing even a small amount of weight can markedly enhance insulin sensitivity, leading to improved glucose metabolism and reduced circulating insulin levels.172 This improvement not only helps restore hormonal balance but also promotes regular menstrual cycles and ovulation, which are often disrupted in women with PCOS.173 Consequently, modest weight loss can improve fertility outcomes while reducing the risk of long-term complications like type 2 diabetes and cardiovascular disease.
In addition to its metabolic benefits, weight loss positively impacts psychological well-being. Women with PCOS often experience depression, anxiety, and body image concerns, which can be alleviated through successful weight management.174 Dietary strategies that emphasize whole foods with a low glycemic index are particularly effective in supporting mood disruptions in people with PCOS.
Physical activity complements weight management efforts by directly addressing many of the underlying mechanisms of PCOS. Exercise improves insulin sensitivity independently of weight loss, making it an essential intervention for women across all body types.175 Furthermore, regular physical activity helps regulate hormone levels by reducing free testosterone and improving symptoms such as acne and hirsutism.176 It also promotes better energy balance by increasing lean muscle mass and metabolic rate.177
Different types of exercise provide unique benefits for women with PCOS:
· Aerobic Exercise: Activities like walking, jogging, cycling, or swimming improve cardiovascular fitness while enhancing insulin sensitivity. Aerobic exercise is particularly effective for reducing visceral fat, which is closely linked to metabolic dysfunction in PCOS.178
· High-Intensity Interval Training (HIIT): HIIT has gained attention for its ability to improve insulin resistance and reduce androgen levels without requiring long workout sessions. This makes it an efficient option for managing both metabolic and hormonal symptoms.179
· Strength Training: Resistance exercises help build muscle mass and reduce body fat while improving metabolic markers such as fasting insulin levels. Increased muscle mass also enhances long-term glucose regulation.180
· Mind-Body Practices: Stress management is critical in PCOS since elevated stress can worsen hormonal imbalances. Exercises like yoga and Pilates not only reduce stress but also improve mental well-being by alleviating symptoms of depression and anxiety.181
The benefits of exercise extend beyond physical health. Regular activity boosts mood through the release of endorphins, enhances sleep quality, and fosters a sense of empowerment, all of which contribute to better management of PCOS symptoms. Importantly, sustained engagement in physical activity is key to achieving long-term improvements.
When combined, modest weight loss, regular exercise, and dietary changes create a synergistic effect that maximizes health outcomes for women with PCOS. Weight loss amplifies the benefits of exercise by further improving insulin sensitivity and reducing inflammation, while exercise enhances weight management efforts by increasing energy expenditure and preserving lean muscle mass during calorie restriction. Together, these interventions address both the reproductive and metabolic complications of PCOS while promoting psychological resilience.
By adopting a holistic approach that integrates sustainable dietary changes with consistent physical activity, women with PCOS can achieve meaningful improvements in their condition. This layered strategy not only alleviates immediate symptoms but also reduces the risk of long-term health complications associated with PCOS.
Who Are the Experts
Below is a list of prominent researchers and clinicians specializing in PCOS, along with their affiliations, expertise, key contributions, and publication information.
1. Mariangela Rondanelli, MD, PHD
a. Affiliation: University of Pavia, Pavia, Italy
b. Expertise: Polycystic Ovary Syndrome
c. Key Work: Berberine Phospholipid Is an Effective Insulin Sensitizer and Improves Metabolic and Hormonal Disorders in Women with Polycystic Ovary Syndrome.
d. Publications:
Rondanelli M, Riva A, Petrangolini G, et al. Berberine phospholipid is an effective insulin sensitizer and improves metabolic and hormonal disorders in women with polycystic ovary syndrome: a one-group pretest–post-test explanatory study. Nutrients. 2021;13(10):3665.
Rondanelli M, Infantino V, Riva A, Petrangolini G, Faliva MA, Peroni G, Naso M, Nichetti M, Spadaccini D, Gasparri C, Perna S. Polycystic ovary syndrome management: a review of the possible amazing role of berberine. Arch Gynecol Obstet. 2020 Jan;301(1):53-60. doi: 10.1007/s00404-020-05450-4. Epub 2020 Feb 14. PMID: 32060683; PMCID: PMC7028834.
e. Contact Info: mariangela.rondanelli@unipv.it
2. Anuja Dokras, MD, MHCI, PhD
a. Affiliation: University of Pennsylvania; Executive Director, Women’s Health Center for Clinical Innovation; Director, Penn PCOS Center.
b. Expertise: Reproductive endocrinology, translational medicine, clinical trials, and multidisciplinary care for PCOS.
c. Key Work: NIH-funded researcher; co-editor of Yen and Jaffe’s Reproductive Endocrinology; President of the Androgen Excess-PCOS Society (2015-2017); extensive work on evidence-based guidelines for PCOS management.
d. Publications:
Teede HJ, Tay CT, Laven JJE, Dokras A, Moran LJ, Piltonen TT, Costello MF, Boivin J, Redman LM, Boyle JA, Norman RJ, Mousa A, Joham AE; International PCOS Network. Recommendations from the 2023 international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Eur J Endocrinol. 2023 Aug 2;189(2):G43-G64. doi: 10.1093/ejendo/lvad096. PMID: 37580861.Dokras A. Does body weight affect cardiometabolic risk in women with polycystic ovary syndrome? Fertil Steril. 2019 Jan;111(1):56-57. doi: 10.1016/j.fertnstert.2018.11.001. PMID: 30611418.
Dokras A, Sarwer DB, Allison KC, Milman L, Kris-Etherton PM, Kunselman AR, Stetter CM, Williams NI, Gnatuk CL, Estes SJ, Fleming J, Coutifaris C, Legro RS. Weight Loss and Lowering Androgens Predict Improvements in Health-Related Quality of Life in Women With PCOS. J Clin Endocrinol Metab. 2016 Aug;101(8):2966-74. doi: 10.1210/jc.2016-1896. Epub 2016 Jun 2. PMID: 27253669; PMCID: PMC4971336.
Dokras A, Saini S, Gibson-Helm M, Schulkin J, Cooney L, Teede H. Gaps in knowledge among physicians regarding diagnostic criteria and management of polycystic ovary syndrome. Fertil Steril. 2017 Jun;107(6):1380-1386.e1. doi: 10.1016/j.fertnstert.2017.04.011. Epub 2017 May 5. PMID: 28483503.
e. Contact Information: adokras@obgyn.upenn.edu
3. Richard S. Legro, MD
a. Affiliation: Penn State College of Medicine; Chair of Obstetrics and Gynecology.
b. Expertise: Diagnosis and treatment of PCOS, infertility trials, genetic/environmental causes of PCOS.
c. Key Work: Lead investigator for NIH-funded Pregnancy in PCOS trials; published over 300 peer-reviewed articles; contributed to diagnostic criteria for PCOS.
d. Publications:
Legro RS, Arslanian SA, Ehrmann DA, Hoeger KM, Murad MH, Pasquali R, Welt CK; Endocrine Society. Diagnosis and treatment of polycystic ovary syndrome: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2013 Dec;98(12):4565-92. doi: 10.1210/jc.2013-2350. Epub 2013 Oct 22. Erratum in: J Clin Endocrinol Metab. 2021 May 13;106(6):e2462. doi: 10.1210/clinem/dgab248. PMID: 24151290; PMCID: PMC5399492.
Legro RS. The genetics of polycystic ovary syndrome. Am J Med. 1995 Jan 16;98(1A):9S-16S. doi: 10.1016/s0002-9343(99)80053-9. PMID: 7825646.Legro RS, Kunselman AR, Dodson WC, Dunaif A. Prevalence and predictors of risk for type 2 diabetes mellitus and impaired glucose tolerance in polycystic ovary syndrome: a prospective, controlled study in 254 affected women. J Clin Endocrinol Metab. 1999 Jan;84(1):165-9. doi: 10.1210/jcem.84.1.5393. PMID: 9920077.
e. Contact Information: rsl1@psu.edu
4. Andrea E. Dunaif, MD
a. Affiliation: Mount Sinai Health System; Chief of Endocrinology, Diabetes and Bone Disease.
b. Expertise: Genetics of PCOS, insulin resistance in PCOS, metabolic disorders.
c. Key Work: Mapping chromosomal regions linked to PCOS genes; established links between PCOS and type 2 diabetes; NIH-funded research on therapeutic targets.
d. Publications:
Exaggerated glucagon responses to hypoglycemia in women with polycystic ovary syndrome. Sam S, Vellanki P, Yalamanchi SK, Bergman RN, Dunaif A Metabolism: Clinical and Experimental. 2017 Jun; 71: 125-131. doi:http://dx.doi.org/10.1016/j.metabol.2017.03.008. PMID: 28521865 PMCID: 28521865 ISSN: 0026-0495
Pathogenic Anti-Müllerian Hormone Variants in Polycystic Ovary Syndrome.
Gorsic LK, Kosova G, Werstein B, Sisk R, Legro RS, Hayes MG, Teixeira JM, Dunaif A, Urbanek M. J Clin Endocrinol Metab. 2017 Aug 1;102(8):2862-2872. doi: 10.1210/jc.2017-00612. PMID:28505284 ISSN PMCID: PMC5546867 ISSN: 0021-972
Polycystic ovary syndrome. Azziz R, Carmina E, Chen Z, Dunaif A, Laven JSE, Legro RS, Lizneva D, Natterson-Horowtiz B, Teede HJ, Yildiz BO. Nature Reviews Disease Primers. 2016 Aug 11; 2: doi:http://dx.doi.org/10.1038/nrdp.2016.57. PMID: 27510637
e. Contact Information: andrea.dunaif@mssm.edu
5. John E. Nestler, MD
a. Affiliation: Virginia Commonwealth University (VCU); Chair of Internal Medicine.
b. Expertise: Insulin resistance in PCOS, infertility treatments using insulin-sensitizing drugs.
c. Key Work: Seminal research establishing insulin-sensitizing drugs as infertility treatments; principal investigator for multiple NIH grants.
d. Publications:
Essah PA, Nestler JE. Metabolic syndrome in women with polycystic ovary syndrome. Fertil Steril. 2006 Jul;86 Suppl 1:S18-9. doi: 10.1016/j.fertnstert.2006.04.013. PMID: 16798277.
Apridonidze T, Essah PA, Iuorno MJ, Nestler JE. Prevalence and characteristics of the metabolic syndrome in women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2005 Apr;90(4):1929-35. doi: 10.1210/jc.2004-1045. Epub 2004 Dec 28. PMID: 15623819.
Nestler JE, Jakubowicz DJ, Reamer P, Gunn RD, Allan G. Ovulatory and metabolic effects of D-chiro-inositol in the polycystic ovary syndrome. N Engl J Med. 1999 Apr 29;340(17):1314-20. doi: 10.1056/NEJM199904293401703. PMID: 10219066.
e. Contact Information: john.nestler@vcuhealth.org
Social Media Contributors
The following list includes a mix of dietitians, nutritionists, coaches, and advocates who are actively sharing information and support for individuals with PCOS on social media platforms, particularly Instagram. Many of these influencers have personal experience with PCOS and use their platforms to raise awareness, provide nutritional advice, and offer support to others with the condition.
1. Tallene Hacatoryan
· Affiliation: The Cysterhood
· Expertise: PCOS Dietitian
· Key work: Helping women lose weight with PCOS and reverse symptoms
· Contact: @pcos.weightloss on Instagram, https://www.youtube.com/channel/UC7-AdIT2AHwo95LdQpJFdVA
· Publications: Blog at pcosweightloss.org
2. Cailee Fischer
· Affiliation: Independent
· Expertise: PCOS nutrition and recipes
· Key work: Sharing simple and healthy recipes for PCOS management
· Contact: cailee@lulucreativemedia.com, @caileeeeats on Instagram
3. Drew Baird
· Affiliation: Health & Balance Vitamins, Novum PCOS
· Expertise: PCOS Coaching
· Key work: PCOS mentoring and supplement recommendations
· Contact: @thepcosmentor on Instagram3
4. Melissa Azzaro
· Affiliation: Independent
· Expertise: Hormones, Metabolism, Peri/Menopause
· Key work: Hormone balancing and metabolism fixing
· Contact: @the.hormone.dietitian on Instagram
5. Sam Abbott
· Affiliation: PCOS Nutrition Co.
· Expertise: PCOS Nutrition
· Key work: Reducing PCOS symptoms without restriction
· Contact: https://pcosnutritionco.com/, hello@pcosnutritionco.com, @pcos.nutritionist on Instagram
6. Clare Goodwin
· Affiliation: Ovie Health
· Expertise: Registered Nutritionist
· Key work: Author of “Getting Pregnant with PCOS”
· Contact: @thepcosnutritionist on Instagram
· Publications: Book “Getting Pregnant with PCOS”, Podcast
7. Caitlin Johnson
· Affiliation: PCOS Formularies
· Expertise: PCOS Dietitian
· Key work: Creator of the PCOS App
· Contact: https://pcosfertilitynutrition.com/, @pcosfertilitynutrition on Instagram
8. Roshni Chandrasekar
· Affiliation: Independent
· Expertise: Stanford Certified Nutritionist
· Key work: One-on-one programs for weight loss, conceiving, and PCOS management
· Contact: @pcos.cysterly_health on Instagram
9. Genevieve Sanders
· Affiliation: Independent
· Expertise: Registered Dietitian Nutritionist, Certified Personal Trainer
· Key work: Helping regulate cycles and thrive with PCOS
· Contact: Email contact@gentherdn.com, @gen.the.pcos.rdn on Instagram
Bibliography
Abdelazeem, Basel, Kirellos Said Abbas, Joseph Shehata, Nischit Baral, Sandi Banour, and Mustafa Hassan. “The Effects of Curcumin as Dietary Supplement for Patients with Polycystic Ovary Syndrome: An Updated Systematic Review and Meta‐analysis of Randomized Clinical Trials.” Phytotherapy Research 36, no. 1 (January 2022): 22–32. https://doi.org/10.1002/ptr.7274.
Aboeldalyl, Shaimaa, Cathryn James, Emaduldin Seyam, Emad Moussa Ibrahim, Hossam El-Din Shawki, and Saad Amer. “The Role of Chronic Inflammation in Polycystic Ovarian Syndrome—A Systematic Review and Meta-Analysis.” International Journal of Molecular Sciences 22, no. 5 (March 8, 2021): 2734. https://doi.org/10.3390/ijms22052734.
Aggarwal, Bharat B., and Kuzhuvelil B. Harikumar. “Potential Therapeutic Effects of Curcumin, the Anti-Inflammatory Agent, against Neurodegenerative, Cardiovascular, Pulmonary, Metabolic, Autoimmune and Neoplastic Diseases.” The International Journal of Biochemistry & Cell Biology 41, no. 1 (January 2009): 40–59. https://doi.org/10.1016/j.biocel.2008.06.010.
Ahmed, Eman M. Sayed, Salem Mohamed E., and Mohamed Samir Eid Sweed. “Effect of Lifestyle Modifications on Polycystic Ovarian Syndrome Symptoms.” Journal of American Science 8, no. 8 (2012): 535–44.
Alahmari, Layla A. “Dietary Fiber Influence on Overall Health, with an Emphasis on CVD, Diabetes, Obesity, Colon Cancer, and Inflammation.” Frontiers in Nutrition 11 (December 13, 2024): 1510564. https://doi.org/10.3389/fnut.2024.1510564.
Alesi, Simon, Carolyn Ee, Lisa J Moran, Vibhuti Rao, and Aya Mousa. “Nutritional Supplements and Complementary Therapies in Polycystic Ovary Syndrome.” Advances in Nutrition 13, no. 4 (July 2022): 1243–66. https://doi.org/10.1093/advances/nmab141.
Anderson, James W, Pat Baird, Richard H Davis Jr, Stefanie Ferreri, Mary Knudtson, Ashraf Koraym, Valerie Waters, and Christine L Williams. “Health Benefits of Dietary Fiber.” Nutrition Reviews 67, no. 4 (April 2009): 188–205. https://doi.org/10.1111/j.1753-4887.2009.00189.x.
Arentz, Susan, Caroline A Smith, Jason Abbott, and Alan Bensoussan. “Diet and Exercise as a Management Strategy for Symptoms of Polycystic Ovary Syndrome (PCOS): The Views of Women in the Community with PCOS.” In Review, April 24, 2020. https://doi.org/10.21203/rs.3.rs-20598/v1.
Asan, Sabiha Ataç, Murat Baş, Berna Eren, and Esen Karaca. “The Effects of Curcumin Supplementation Added to Diet on Anthropometric and Biochemical Status in Women with Polycystic Ovary Syndrome: A Randomized, Placebo-Controlled Trial.” Progress in Nutrition 22, no. 4 (January 11, 2021): e2020089. https://doi.org/10.23751/pn.v22i4.10460.
Ashika Ajitkumar, Ashika Ajitkumar, Gaurav Mohan, Medha Ghose, Sivanaga Yarrarapu, and Swara Afiniwala. “Drug-Induced Liver Injury Secondary to Turmeric Use.” European Journal of Case Reports in Internal Medicine, April 21, 2023. https://doi.org/10.12890/2023_003845.
Azziz, Ricardo, Enrico Carmina, Didier Dewailly, Evanthia Diamanti-Kandarakis, Hector F. Escobar-Morreale, Walter Futterweit, Onno E. Janssen, et al. “Criteria for Defining Polycystic Ovary Syndrome as a Predominantly Hyperandrogenic Syndrome: An Androgen Excess Society Guideline.” The Journal of Clinical Endocrinology & Metabolism 91, no. 11 (November 1, 2006): 4237–45. https://doi.org/10.1210/jc.2006-0178.
Bansal, Anu, Jasobanta Sethi, and Raju K. Parasher. “Role of Structured Exercise Programs in Polycystic Ovary Syndrome: A Systematic Review.” Physical Therapy Reviews 28, no. 1 (January 2, 2023): 49–59. https://doi.org/10.1080/10833196.2022.2163076.
Buckley, Mitchell S, Angela D Goff, and Walter E Knapp. “Fish Oil Interaction with Warfarin.” Annals of Pharmacotherapy 38, no. 1 (January 2004): 50–53. https://doi.org/10.1345/aph.1D007.
Buhrmann, Constanze, Ali Mobasheri, Franziska Busch, Constance Aldinger, Ralf Stahlmann, Azadeh Montaseri, and Mehdi Shakibaei. “Curcumin Modulates Nuclear Factor κB (NF-κB)-Mediated Inflammation in Human Tenocytes in Vitro.” Journal of Biological Chemistry 286, no. 32 (August 2011): 28556–66. https://doi.org/10.1074/jbc.M111.256180.
Calder, Philip C. “Omega-3 Fatty Acids and Inflammatory Processes.” Nutrients 2, no. 3 (March 18, 2010): 355–74. https://doi.org/10.3390/nu2030355.
———. “Omega‐3 Polyunsaturated Fatty Acids and Inflammatory Processes: Nutrition or Pharmacology?” British Journal of Clinical Pharmacology 75, no. 3 (March 2013): 645–62. https://doi.org/10.1111/j.1365-2125.2012.04374.x.
Chien, Yung-Jiun, Chun-Yu Chang, Meng-Yu Wu, Chih-Hao Chen, Yi-Shiung Horng, and Hsin-Chi Wu. “Effects of Curcumin on Glycemic Control and Lipid Profile in Polycystic Ovary Syndrome: Systematic Review with Meta-Analysis and Trial Sequential Analysis.” Nutrients 13, no. 2 (February 21, 2021): 684. https://doi.org/10.3390/nu13020684.
Contreras, Francisca, Werd Al-Najim, and Carel W. Le Roux. “Health Benefits Beyond the Scale: The Role of Diet and Nutrition During Weight Loss Programmes.” Nutrients 16, no. 21 (October 22, 2024): 3585. https://doi.org/10.3390/nu16213585.
Cutler, Dylan A., Sheila M. Pride, and Anthony P. Cheung. “Low Intakes of Dietary Fiber and Magnesium Are Associated with Insulin Resistance and Hyperandrogenism in Polycystic Ovary Syndrome: A Cohort Study.” Food Science & Nutrition 7, no. 4 (April 2019): 1426–37. https://doi.org/10.1002/fsn3.977.
Dennett, Carrie C., and Judy Simon. “The Role of Polycystic Ovary Syndrome in Reproductive and Metabolic Health: Overview and Approaches for Treatment.” Diabetes Spectrum 28, no. 2 (May 1, 2015): 116–20. https://doi.org/10.2337/diaspect.28.2.116.
Di Lorenzo, M., N. Cacciapuoti, M. S. Lonardo, G. Nasti, C. Gautiero, A. Belfiore, B. Guida, and M. Chiurazzi. “Pathophysiology and Nutritional Approaches in Polycystic Ovary Syndrome (PCOS): A Comprehensive Review.” Current Nutrition Reports 12, no. 3 (May 22, 2023): 527–44. https://doi.org/10.1007/s13668-023-00479-8.
El-Saadony, Mohamed T., Tao Yang, Sameh A. Korma, Mahmoud Sitohy, Taia A. Abd El-Mageed, Samy Selim, Soad K. Al Jaouni, et al. “Impacts of Turmeric and Its Principal Bioactive Curcumin on Human Health: Pharmaceutical, Medicinal, and Food Applications: A Comprehensive Review.” Frontiers in Nutrition 9 (January 10, 2023): 1040259. https://doi.org/10.3389/fnut.2022.1040259.
Ferreira, Inês, Filipa Falcato, Narcisa Bandarra, and Amélia P. Rauter. “Resolvins, Protectins, and Maresins: DHA-Derived Specialized Pro-Resolving Mediators, Biosynthetic Pathways, Synthetic Approaches, and Their Role in Inflammation.” Molecules 27, no. 5 (March 3, 2022): 1677. https://doi.org/10.3390/molecules27051677.
Filardi, Tiziana, Rosaria Varì, Elisabetta Ferretti, Alessandra Zicari, Susanna Morano, and Carmela Santangelo. “Curcumin: Could This Compound Be Useful in Pregnancy and Pregnancy-Related Complications?” Nutrients 12, no. 10 (October 17, 2020): 3179. https://doi.org/10.3390/nu12103179.
Galyean, M.L., and M.E. Hubbert. “REVIEW: Traditional and Alternative Sources of Fiber—Roughage Values, Effectiveness, and Levels in Starting and Finishing Diets.” The Professional Animal Scientist 30, no. 6 (December 2014): 571–84. https://doi.org/10.15232/pas.2014-01329.
GHorbani, Zeinab, Azita Hekmatdoost, and Parvin Mirmiran. “Anti-Hyperglycemic and Insulin Sensitizer Effects of Turmeric and Its Principle Constituent Curcumin.” International Journal of Endocrinology and Metabolism 12, no. 4 (October 1, 2014). https://doi.org/10.5812/ijem.18081.
Grygiel-Górniak, Bogna. “Peroxisome Proliferator-Activated Receptors and Their Ligands: Nutritional and Clinical Implications – a Review.” Nutrition Journal 13, no. 1 (December 2014): 17. https://doi.org/10.1186/1475-2891-13-17.
Harrison, C. L., C. B. Lombard, L. J. Moran, and H. J. Teede. “Exercise Therapy in Polycystic Ovary Syndrome: A Systematic Review.” Human Reproduction Update 17, no. 2 (March 1, 2011): 171–83. https://doi.org/10.1093/humupd/dmq045.
Hazlehurst, Jonathan M., Pushpa Singh, Gurkiran Bhogal, Sophie Broughton, and Abd A. Tahrani. “How to Manage Weight Loss in Women with Obesity and PCOS Seeking Fertility?” Clinical Endocrinology 97, no. 2 (August 2022): 208–16. https://doi.org/10.1111/cen.14726.
Heshmati, Javad, Ashraf Moini, Mahdi Sepidarkish, Mojgan Morvaridzadeh, Masoud Salehi, Andriko Palmowski, Maryam Farid Mojtahedi, and Farzad Shidfar. “Effects of Curcumin Supplementation on Blood Glucose, Insulin Resistance and Androgens in Patients with Polycystic Ovary Syndrome: A Randomized Double-Blind Placebo-Controlled Clinical Trial.” Phytomedicine 80 (January 2021): 153395. https://doi.org/10.1016/j.phymed.2020.153395.
Himelein, Melissa J., and Samuel S. Thatcher. “Polycystic Ovary Syndrome and Mental Health: A Review:” Obstetrical & Gynecological Survey 61, no. 11 (November 2006): 723–32. https://doi.org/10.1097/01.ogx.0000243772.33357.84.
Holscher, Hannah D. “Dietary Fiber and Prebiotics and the Gastrointestinal Microbiota.” Gut Microbes 8, no. 2 (March 4, 2017): 172–84. https://doi.org/10.1080/19490976.2017.1290756.
Ioniță-Mîndrican, Corina-Bianca, Khaled Ziani, Magdalena Mititelu, Eliza Oprea, Sorinel Marius Neacșu, Elena Moroșan, Denisa-Elena Dumitrescu, Adrian Cosmin Roșca, Doina Drăgănescu, and Carolina Negrei. “Therapeutic Benefits and Dietary Restrictions of Fiber Intake: A State of the Art Review.” Nutrients 14, no. 13 (June 26, 2022): 2641. https://doi.org/10.3390/nu14132641.
Jakubczyk, Karolina, Aleksandra Drużga, Janda Katarzyna, and Karolina Skonieczna-Żydecka. “Antioxidant Potential of Curcumin—A Meta-Analysis of Randomized Clinical Trials.” Antioxidants 9, no. 11 (November 6, 2020): 1092. https://doi.org/10.3390/antiox9111092.
Jurczewska, Justyna, Joanna Ostrowska, Magdalena Chełchowska, Mariusz Panczyk, Ewa Rudnicka, Marek Kucharski, Roman Smolarczyk, and Dorota Szostak-Węgierek. “Physical Activity, Rather Than Diet, Is Linked to Lower Insulin Resistance in PCOS Women—A Case-Control Study.” Nutrients 15, no. 9 (April 27, 2023): 2111. https://doi.org/10.3390/nu15092111.
Kamal, Datu Agasi Mohd, Norizam Salamt, Allia Najmie Muhammad Yusuf, Mohd Izhar Ariff Mohd Kashim, and Mohd Helmy Mokhtar. “Potential Health Benefits of Curcumin on Female Reproductive Disorders: A Review.” Nutrients 13, no. 9 (September 7, 2021): 3126. https://doi.org/10.3390/nu13093126.
Kar, Avijit, Pratik Ghosh, Prasanta Patra, Deep Sankar Chini, Ashim Kumar Nath, Jayanta Kumar Saha, and Bidhan Chandra Patra. “Omega-3 Fatty Acids Mediated Cellular Signaling and Its Regulation in Human Health.” Clinical Nutrition Open Science 52 (December 2023): 72–86. https://doi.org/10.1016/j.nutos.2023.10.004.
Khalid, Waseem, Muhammad Sajid Arshad, Ayesha Jabeen, Faqir Muhammad Anjum, Tahira Batool Qaisrani, and Hafiz Ansar Rasul Suleria. “Fiber‐enriched Botanicals: A Therapeutic Tool against Certain Metabolic Ailments.” Food Science & Nutrition 10, no. 10 (October 2022): 3203–18. https://doi.org/10.1002/fsn3.2920.
Khani, Behnaz, Farahnaz Mardanian, and SajadehJafari Fesharaki. “Omega-3 Supplementation Effects on Polycystic Ovary Syndrome Symptoms and Metabolic Syndrome.” Journal of Research in Medical Sciences 22, no. 1 (2017): 64. https://doi.org/10.4103/jrms.JRMS_644_16.
Komal, Fiza, Muhammad Kamran Khan, Muhammad Imran, Muhammad Haseeb Ahmad, Haseeb Anwar, Usman Ali Ashfaq, Nazir Ahmad, et al. “Impact of Different Omega-3 Fatty Acid Sources on Lipid, Hormonal, Blood Glucose, Weight Gain and Histopathological Damages Profile in PCOS Rat Model.” Journal of Translational Medicine 18, no. 1 (December 2020): 349. https://doi.org/10.1186/s12967-020-02519-1.
Krupa, Kristina N., Kristina Fritz, and Mayur Parmar. “Omega-3 Fatty Acids.” In StatPearls. Treasure Island (FL): StatPearls Publishing, 2025. http://www.ncbi.nlm.nih.gov/books/NBK564314/.
Lancaster, Samuel M., Brittany Lee-McMullen, Charles Wilbur Abbott, Jeniffer V. Quijada, Daniel Hornburg, Kévin Contrepois, Chia-Jui Hung, et al. “Global, Distinctive, and Personal Changes in Molecular and Microbial Profiles by Specific Fibers in Humans.” Cell Host & Microbe 30, no. 6 (June 8, 2022): 848–62.
Lattimer, James M., and Mark D. Haub. “Effects of Dietary Fiber and Its Components on Metabolic Health.” Nutrients 2, no. 12 (December 15, 2010): 1266–89. https://doi.org/10.3390/nu2121266.
Legro, Richard S. “Polycystic Ovary Syndrome and Cardiovascular Disease: A Premature Association?” Endocrine Reviews 24, no. 3 (June 1, 2003): 302–12. https://doi.org/10.1210/er.2003-0004.
Leung, Wing Ting, Zhijing Tang, Yuanyuan Feng, Haiyun Guan, Zengshu Huang, and Wei Zhang. “Lower Fiber Consumption in Women with Polycystic Ovary Syndrome: A Meta-Analysis of Observational Studies.” Nutrients 14, no. 24 (December 12, 2022): 5285. https://doi.org/10.3390/nu14245285.
Liao, Wan-Ting, Jing-Yang Huang, Ming-Tsung Lee, Yu-Cih Yang, and Chun-Chi Wu. “Higher Risk of Type 2 Diabetes in Young Women with Polycystic Ovary Syndrome: A 10-Year Retrospective Cohort Study.” World Journal of Diabetes 13, no. 3 (March 15, 2022): 240–50. https://doi.org/10.4239/wjd.v13.i3.240.
Lim, Siew S, Peter M Clifton, Manny Noakes, and Robert J Norman. “Obesity Management in Women with Polycystic Ovary Syndrome.” Women’s Health 3, no. 1 (January 2007): 73–86. https://doi.org/10.2217/17455057.3.1.73.
Maarouf, Taiseer, Doaa Mohamed, Aalaa Tantawy, and Passant Eid. “Effect of Omega-3 Fatty Acids on Hormonal Profile and Ovarian Stromal Blood Flow in Patients with Polycystic Ovary Syndrome.” Evidence Based Women’s Health Journal 9, no. 4 (November 1, 2019): 542–48. https://doi.org/10.21608/ebwhj.2019.64357.
Madison, Annelise, and Janice K Kiecolt-Glaser. “Stress, Depression, Diet, and the Gut Microbiota: Human–Bacteria Interactions at the Core of Psychoneuroimmunology and Nutrition.” Current Opinion in Behavioral Sciences 28 (August 2019): 105–10. https://doi.org/10.1016/j.cobeha.2019.01.011.
Manta, Aspasia, Stavroula Paschou, Georgia Isari, Ioanna Mavroeidi, Sophia Kalantaridou, and Melpomeni Peppa. “Glycemic Index and Glycemic Load Estimates in the Dietary Approach of Polycystic Ovary Syndrome.” Nutrients 15, no. 15 (August 7, 2023): 3483. https://doi.org/10.3390/nu15153483.
Marshall, John C., and Andrea Dunaif. “Should All Women with PCOS Be Treated for Insulin Resistance?” Fertility and Sterility 97, no. 1 (January 2012): 18–22. https://doi.org/10.1016/j.fertnstert.2011.11.036.
McRorie, Johnson W. “Evidence-Based Approach to Fiber Supplements and Clinically Meaningful Health Benefits, Part 2: What to Look for and How to Recommend an Effective Fiber Therapy.” Nutrition Today 50, no. 2 (March 2015): 90–97. https://doi.org/10.1097/NT.0000000000000089.
Mohammadi, Elahe, and Maryam Rafraf. “Benefits of Omega-3 Fatty Acids Supplementation on Serum Paraoxonase 1 Activity and Lipids Ratios in Polycystic Ovary Syndrome.” Text/html. Health Promotion Perspectives; ISSN: 2228-6497, 2012. https://doi.org/10.5681/HPP.2012.023.
Murga-Garrido, Sofia M., Qilin Hong, Tzu-Wen L. Cross, Evan R. Hutchison, Jessica Han, Sydney P. Thomas, Eugenio I. Vivas, et al. “Gut Microbiome Variation Modulates the Effects of Dietary Fiber on Host Metabolism.” Microbiome 9, no. 1 (December 2021): 117. https://doi.org/10.1186/s40168-021-01061-6.
Norman, Robert J, Didier Dewailly, Richard S Legro, and Theresa E Hickey. “Polycystic Ovary Syndrome.” The Lancet 370, no. 9588 (August 2007): 685–97. https://doi.org/10.1016/S0140-6736(07)61345-2.
“Office of Dietary Supplements – Omega-3 Fatty Acids.” Accessed November 25, 2024. https://ods.od.nih.gov/factsheets/Omega3FattyAcids-Consumer/.
“Polycystic Ovary Syndrome.” Accessed February 18, 2025. https://www.who.int/news-room/fact-sheets/detail/polycystic-ovary-syndrome.
Prasad, Sahdeo, Amit K. Tyagi, and Bharat B. Aggarwal. “Recent Developments in Delivery, Bioavailability, Absorption and Metabolism of Curcumin: The Golden Pigment from Golden Spice.” Cancer Research and Treatment 46, no. 1 (January 15, 2014): 2–18. https://doi.org/10.4143/crt.2014.46.1.2.
Purwar, Ananya, and Shailesh Nagpure. “Insulin Resistance in Polycystic Ovarian Syndrome.” Cureus, October 16, 2022. https://doi.org/10.7759/cureus.30351.
Rojas, Joselyn, Mervin Chávez, Luis Olivar, Milagros Rojas, Jessenia Morillo, José Mejías, María Calvo, and Valmore Bermúdez. “Polycystic Ovary Syndrome, Insulin Resistance, and Obesity: Navigating the Pathophysiologic Labyrinth.” International Journal of Reproductive Medicine 2014 (2014): 1–17. https://doi.org/10.1155/2014/719050.
Saadati, Najmieh, Fatemeh Haidari, Mojgan Barati, Roshan Nikbakht, Golshan Mirmomeni, and Fakher Rahim. “The Effect of Low Glycemic Index Diet on the Reproductive and Clinical Profile in Women with Polycystic Ovarian Syndrome: A Systematic Review and Meta-Analysis.” Heliyon 7, no. 11 (November 2021): e08338. https://doi.org/10.1016/j.heliyon.2021.e08338.
Scarfò, Giorgia, Simona Daniele, Jonathan Fusi, Marco Gesi, Claudia Martini, Ferdinando Franzoni, Vito Cela, and Paolo Giovanni Artini. “Metabolic and Molecular Mechanisms of Diet and Physical Exercise in the Management of Polycystic Ovarian Syndrome.” Biomedicines 10, no. 6 (June 2, 2022): 1305. https://doi.org/10.3390/biomedicines10061305.
Singh, Samradhi, Namrata Pal, Swasti Shubham, Devojit Kumar Sarma, Vinod Verma, Francesco Marotta, and Manoj Kumar. “Polycystic Ovary Syndrome: Etiology, Current Management, and Future Therapeutics.” Journal of Clinical Medicine 12, no. 4 (February 11, 2023): 1454. https://doi.org/10.3390/jcm12041454.
Slavin, Joanne. “Fiber and Prebiotics: Mechanisms and Health Benefits.” Nutrients 5, no. 4 (April 22, 2013): 1417–35. https://doi.org/10.3390/nu5041417.
Szczuko, Małgorzata, Justyna Kikut, Urszula Szczuko, Iwona Szydłowska, Jolanta Nawrocka-Rutkowska, Maciej Ziętek, Donatella Verbanac, and Luciano Saso. “Nutrition Strategy and Life Style in Polycystic Ovary Syndrome—Narrative Review.” Nutrients 13, no. 7 (July 18, 2021): 2452. https://doi.org/10.3390/nu13072452.
Wang, Erica T., Ronit Calderon-Margalit, Marcelle I. Cedars, Martha L. Daviglus, Sharon S. Merkin, Pamela J. Schreiner, Barbara Sternfeld, et al. “Polycystic Ovary Syndrome and Risk for Long-Term Diabetes and Dyslipidemia.” Obstetrics & Gynecology 117, no. 1 (January 2011): 6–13. https://doi.org/10.1097/AOG.0b013e31820209bb.
Wang, Kexin, Yanhua Li, and Yu Chen. “Androgen Excess: A Hallmark of Polycystic Ovary Syndrome.” Frontiers in Endocrinology 14 (December 13, 2023): 1273542. https://doi.org/10.3389/fendo.2023.1273542.
Wang, Xuejiao, Ting Xu, Rui Liu, Guojun Wu, Liping Gu, Yahui Zhang, Feng Zhang, et al. “High-Fiber Diet or Combined With Acarbose Alleviates Heterogeneous Phenotypes of Polycystic Ovary Syndrome by Regulating Gut Microbiota.” Frontiers in Endocrinology 12 (February 2, 2022): 806331. https://doi.org/10.3389/fendo.2021.806331.
White, Nicole. “A Guide to Recommending Fiber Supplements for Self-Care.” American Journal of Lifestyle Medicine 14, no. 6 (November 2020): 589–91. https://doi.org/10.1177/1559827620947375.
Yang, Kailin, Liuting Zeng, Tingting Bao, and Jinwen Ge. “Effectiveness of Omega-3 Fatty Acid for Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis.” Reproductive Biology and Endocrinology 16, no. 1 (December 2018): 27. https://doi.org/10.1186/s12958-018-0346-x.
———. “Effectiveness of Omega-3 Fatty Acid for Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis.” Reproductive Biology and Endocrinology 16, no. 1 (December 2018): 27. https://doi.org/10.1186/s12958-018-0346-x.
Yang, Qin, Qin Wan, and Zhen Wang. “Curcumin Mitigates Polycystic Ovary Syndrome in Mice by Suppressing TLR4/MyD88/NF-κB Signaling Pathway Activation and Reducing Intestinal Mucosal Permeability.” Scientific Reports 14, no. 1 (December 2, 2024): 29848. https://doi.org/10.1038/s41598-024-81034-5.
Zhang, Wei, Cong Peng, Lei Xu, Yutai Zhao, Chaolin Huang, and Ling Lu. “The Therapeutic Effects of Curcumin on Polycystic Ovary Syndrome by Upregulating PPAR-γ Expression and Reducing Oxidative Stress in a Rat Model.” Frontiers in Endocrinology 15 (November 20, 2024): 1494852. https://doi.org/10.3389/fendo.2024.1494852.
“Polycystic Ovary Syndrome,” accessed February 18, 2025, https://www.who.int/news-room/fact-sheets/detail/polycystic-ovary-syndrome.↩︎
Ricardo Azziz et al., “Criteria for Defining Polycystic Ovary Syndrome as a Predominantly Hyperandrogenic Syndrome: An Androgen Excess Society Guideline,” The Journal of Clinical Endocrinology & Metabolism 91, no. 11 (November 1, 2006): 4237–45, https://doi.org/10.1210/jc.2006-0178.↩︎
Shaimaa Aboeldalyl et al., “The Role of Chronic Inflammation in Polycystic Ovarian Syndrome—A Systematic Review and Meta-Analysis,” International Journal of Molecular Sciences 22, no. 5 (March 8, 2021): 2734, https://doi.org/10.3390/ijms22052734.↩︎
Małgorzata Szczuko et al., “Nutrition Strategy and Life Style in Polycystic Ovary Syndrome—Narrative Review,” Nutrients 13, no. 7 (July 18, 2021): 2452, https://doi.org/10.3390/nu13072452.↩︎
Francisca Contreras, Werd Al-Najim, and Carel W. Le Roux, “Health Benefits Beyond the Scale: The Role of Diet and Nutrition During Weight Loss Programmes,” Nutrients 16, no. 21 (October 22, 2024): 3585, https://doi.org/10.3390/nu16213585.↩︎
Justyna Jurczewska et al., “Physical Activity, Rather Than Diet, Is Linked to Lower Insulin Resistance in PCOS Women—A Case-Control Study,” Nutrients 15, no. 9 (April 27, 2023): 2111, https://doi.org/10.3390/nu15092111.↩︎
Aboeldalyl et al., “The Role of Chronic Inflammation in Polycystic Ovarian Syndrome—A Systematic Review and Meta-Analysis.”↩︎
Wan-Ting Liao et al., “Higher Risk of Type 2 Diabetes in Young Women with Polycystic Ovary Syndrome: A 10-Year Retrospective Cohort Study,” World Journal of Diabetes 13, no. 3 (March 15, 2022): 240–50, https://doi.org/10.4239/wjd.v13.i3.240.↩︎
Aboeldalyl et al., “The Role of Chronic Inflammation in Polycystic Ovarian Syndrome—A Systematic Review and Meta-Analysis.”↩︎
Richard S. Legro, “Polycystic Ovary Syndrome and Cardiovascular Disease: A Premature Association?,” Endocrine Reviews 24, no. 3 (June 1, 2003): 302–12, https://doi.org/10.1210/er.2003-0004.↩︎
“Polycystic Ovary Syndrome”; Carrie C. Dennett and Judy Simon, “The Role of Polycystic Ovary Syndrome in Reproductive and Metabolic Health: Overview and Approaches for Treatment,” Diabetes Spectrum 28, no. 2 (May 1, 2015): 116–20, https://doi.org/10.2337/diaspect.28.2.116.↩︎
Melissa J. Himelein and Samuel S. Thatcher, “Polycystic Ovary Syndrome and Mental Health: A Review:,” Obstetrical & Gynecological Survey 61, no. 11 (November 2006): 723–32, https://doi.org/10.1097/01.ogx.0000243772.33357.84.↩︎
Szczuko et al., “Nutrition Strategy and Life Style in Polycystic Ovary Syndrome—Narrative Review.”↩︎
Jonathan M. Hazlehurst et al., “How to Manage Weight Loss in Women with Obesity and PCOS Seeking Fertility?,” Clinical Endocrinology 97, no. 2 (August 2022): 208–16, https://doi.org/10.1111/cen.14726.↩︎
Contreras, Al-Najim, and Le Roux, “Health Benefits Beyond the Scale.”↩︎
Robert J Norman et al., “Polycystic Ovary Syndrome,” The Lancet 370, no. 9588 (August 2007): 685–97, https://doi.org/10.1016/S0140-6736(07)61345-2.↩︎
Erica T. Wang et al., “Polycystic Ovary Syndrome and Risk for Long-Term Diabetes and Dyslipidemia,” Obstetrics & Gynecology 117, no. 1 (January 2011): 6–13, https://doi.org/10.1097/AOG.0b013e31820209bb; Samradhi Singh et al., “Polycystic Ovary Syndrome: Etiology, Current Management, and Future Therapeutics,” Journal of Clinical Medicine 12, no. 4 (February 11, 2023): 1454, https://doi.org/10.3390/jcm12041454.↩︎
Kexin Wang, Yanhua Li, and Yu Chen, “Androgen Excess: A Hallmark of Polycystic Ovary Syndrome,” Frontiers in Endocrinology 14 (December 13, 2023): 1273542, https://doi.org/10.3389/fendo.2023.1273542; Singh et al., “Polycystic Ovary Syndrome.”↩︎
Singh et al., “Polycystic Ovary Syndrome.”↩︎
Singh et al.; John C. Marshall and Andrea Dunaif, “Should All Women with PCOS Be Treated for Insulin Resistance?,” Fertility and Sterility 97, no. 1 (January 2012): 18–22, https://doi.org/10.1016/j.fertnstert.2011.11.036.↩︎
Ananya Purwar and Shailesh Nagpure, “Insulin Resistance in Polycystic Ovarian Syndrome,” Cureus, October 16, 2022, https://doi.org/10.7759/cureus.30351.↩︎
Wang et al., “Polycystic Ovary Syndrome and Risk for Long-Term Diabetes and Dyslipidemia.”↩︎
Wang et al.↩︎
Contreras, Al-Najim, and Le Roux, “Health Benefits Beyond the Scale.”↩︎
Szczuko et al., “Nutrition Strategy and Life Style in Polycystic Ovary Syndrome—Narrative Review.”↩︎
M. Di Lorenzo et al., “Pathophysiology and Nutritional Approaches in Polycystic Ovary Syndrome (PCOS): A Comprehensive Review,” Current Nutrition Reports 12, no. 3 (May 22, 2023): 527–44, https://doi.org/10.1007/s13668-023-00479-8.↩︎
C. L. Harrison et al., “Exercise Therapy in Polycystic Ovary Syndrome: A Systematic Review,” Human Reproduction Update 17, no. 2 (March 1, 2011): 171–83, https://doi.org/10.1093/humupd/dmq045; Anu Bansal, Jasobanta Sethi, and Raju K. Parasher, “Role of Structured Exercise Programs in Polycystic Ovary Syndrome: A Systematic Review,” Physical Therapy Reviews 28, no. 1 (January 2, 2023): 49–59, https://doi.org/10.1080/10833196.2022.2163076; Eman M. Sayed Ahmed, Salem Mohamed E., and Mohamed Samir Eid Sweed, “Effect of Lifestyle Modifications on Polycystic Ovarian Syndrome Symptoms,” Journal of American Science 8, no. 8 (2012): 535–44.↩︎
Giorgia Scarfò et al., “Metabolic and Molecular Mechanisms of Diet and Physical Exercise in the Management of Polycystic Ovarian Syndrome,” Biomedicines 10, no. 6 (June 2, 2022): 1305, https://doi.org/10.3390/biomedicines10061305.↩︎
Liao et al., “Higher Risk of Type 2 Diabetes in Young Women with Polycystic Ovary Syndrome.”↩︎
Legro, “Polycystic Ovary Syndrome and Cardiovascular Disease.”↩︎
Aspasia Manta et al., “Glycemic Index and Glycemic Load Estimates in the Dietary Approach of Polycystic Ovary Syndrome,” Nutrients 15, no. 15 (August 7, 2023): 3483, https://doi.org/10.3390/nu15153483.↩︎
Simon Alesi et al., “Nutritional Supplements and Complementary Therapies in Polycystic Ovary Syndrome,” Advances in Nutrition 13, no. 4 (July 2022): 1243–66, https://doi.org/10.1093/advances/nmab141.↩︎
Susan Arentz et al., “Diet and Exercise as a Management Strategy for Symptoms of Polycystic Ovary Syndrome (PCOS): The Views of Women in the Community with PCOS.” (In Review, April 24, 2020), https://doi.org/10.21203/rs.3.rs-20598/v1.↩︎
Najmieh Saadati et al., “The Effect of Low Glycemic Index Diet on the Reproductive and Clinical Profile in Women with Polycystic Ovarian Syndrome: A Systematic Review and Meta-Analysis,” Heliyon 7, no. 11 (November 2021): e08338, https://doi.org/10.1016/j.heliyon.2021.e08338; Manta et al., “Glycemic Index and Glycemic Load Estimates in the Dietary Approach of Polycystic Ovary Syndrome.”↩︎
Di Lorenzo et al., “Pathophysiology and Nutritional Approaches in Polycystic Ovary Syndrome (PCOS).”↩︎
Alesi et al., “Nutritional Supplements and Complementary Therapies in Polycystic Ovary Syndrome.”↩︎
M.L. Galyean and M.E. Hubbert, “REVIEW: Traditional and Alternative Sources of Fiber—Roughage Values, Effectiveness, and Levels in Starting and Finishing Diets,” The Professional Animal Scientist 30, no. 6 (December 2014): 571–84, https://doi.org/10.15232/pas.2014-01329.↩︎
Corina-Bianca Ioniță-Mîndrican et al., “Therapeutic Benefits and Dietary Restrictions of Fiber Intake: A State of the Art Review,” Nutrients 14, no. 13 (June 26, 2022): 2641, https://doi.org/10.3390/nu14132641.↩︎
Ioniță-Mîndrican et al.↩︎
Ioniță-Mîndrican et al.↩︎
Xuejiao Wang et al., “High-Fiber Diet or Combined With Acarbose Alleviates Heterogeneous Phenotypes of Polycystic Ovary Syndrome by Regulating Gut Microbiota,” Frontiers in Endocrinology 12 (February 2, 2022): 806331, https://doi.org/10.3389/fendo.2021.806331; Joselyn Rojas et al., “Polycystic Ovary Syndrome, Insulin Resistance, and Obesity: Navigating the Pathophysiologic Labyrinth,” International Journal of Reproductive Medicine 2014 (2014): 1–17, https://doi.org/10.1155/2014/719050.↩︎
Rojas et al., “Polycystic Ovary Syndrome, Insulin Resistance, and Obesity.”↩︎
Annelise Madison and Janice K Kiecolt-Glaser, “Stress, Depression, Diet, and the Gut Microbiota: Human–Bacteria Interactions at the Core of Psychoneuroimmunology and Nutrition,” Current Opinion in Behavioral Sciences 28 (August 2019): 105–10, https://doi.org/10.1016/j.cobeha.2019.01.011.↩︎
Layla A. Alahmari, “Dietary Fiber Influence on Overall Health, with an Emphasis on CVD, Diabetes, Obesity, Colon Cancer, and Inflammation,” Frontiers in Nutrition 11 (December 13, 2024): 1510564, https://doi.org/10.3389/fnut.2024.1510564.↩︎
Waseem Khalid et al., “Fiber‐enriched Botanicals: A Therapeutic Tool against Certain Metabolic Ailments,” Food Science & Nutrition 10, no. 10 (October 2022): 3203–18, https://doi.org/10.1002/fsn3.2920.↩︎
Joanne Slavin, “Fiber and Prebiotics: Mechanisms and Health Benefits,” Nutrients 5, no. 4 (April 22, 2013): 1417–35, https://doi.org/10.3390/nu5041417; Nicole White, “A Guide to Recommending Fiber Supplements for Self-Care,” American Journal of Lifestyle Medicine 14, no. 6 (November 2020): 589–91, https://doi.org/10.1177/1559827620947375.↩︎
White, “A Guide to Recommending Fiber Supplements for Self-Care.”↩︎
White; James M. Lattimer and Mark D. Haub, “Effects of Dietary Fiber and Its Components on Metabolic Health,” Nutrients 2, no. 12 (December 15, 2010): 1266–89, https://doi.org/10.3390/nu2121266.↩︎
Lattimer and Haub, “Effects of Dietary Fiber and Its Components on Metabolic Health.”↩︎
Hannah D. Holscher, “Dietary Fiber and Prebiotics and the Gastrointestinal Microbiota,” Gut Microbes 8, no. 2 (March 4, 2017): 172–84, https://doi.org/10.1080/19490976.2017.1290756.↩︎
Dylan A. Cutler, Sheila M. Pride, and Anthony P. Cheung, “Low Intakes of Dietary Fiber and Magnesium Are Associated with Insulin Resistance and Hyperandrogenism in Polycystic Ovary Syndrome: A Cohort Study,” Food Science & Nutrition 7, no. 4 (April 2019): 1426–37, https://doi.org/10.1002/fsn3.977.↩︎
Wang et al., “High-Fiber Diet or Combined With Acarbose Alleviates Heterogeneous Phenotypes of Polycystic Ovary Syndrome by Regulating Gut Microbiota.”↩︎
Wing Ting Leung et al., “Lower Fiber Consumption in Women with Polycystic Ovary Syndrome: A Meta-Analysis of Observational Studies,” Nutrients 14, no. 24 (December 12, 2022): 5285, https://doi.org/10.3390/nu14245285.↩︎
Leung et al.↩︎
James W Anderson et al., “Health Benefits of Dietary Fiber,” Nutrition Reviews 67, no. 4 (April 2009): 188–205, https://doi.org/10.1111/j.1753-4887.2009.00189.x.↩︎
White, “A Guide to Recommending Fiber Supplements for Self-Care.”↩︎
Johnson W. McRorie, “Evidence-Based Approach to Fiber Supplements and Clinically Meaningful Health Benefits, Part 2: What to Look for and How to Recommend an Effective Fiber Therapy,” Nutrition Today 50, no. 2 (March 2015): 90–97, https://doi.org/10.1097/NT.0000000000000089.↩︎
Samuel M. Lancaster et al., “Global, Distinctive, and Personal Changes in Molecular and Microbial Profiles by Specific Fibers in Humans,” Cell Host & Microbe 30, no. 6 (June 8, 2022): 848–62.↩︎
Sofia M. Murga-Garrido et al., “Gut Microbiome Variation Modulates the Effects of Dietary Fiber on Host Metabolism,” Microbiome 9, no. 1 (December 2021): 117, https://doi.org/10.1186/s40168-021-01061-6.↩︎
Cutler, Pride, and Cheung, “Low Intakes of Dietary Fiber and Magnesium Are Associated with Insulin Resistance and Hyperandrogenism in Polycystic Ovary Syndrome.”↩︎
White, “A Guide to Recommending Fiber Supplements for Self-Care.”↩︎
Siew S Lim et al., “Obesity Management in Women with Polycystic Ovary Syndrome,” Women’s Health 3, no. 1 (January 2007): 73–86, https://doi.org/10.2217/17455057.3.1.73.↩︎
Taiseer Maarouf et al., “Effect of Omega-3 Fatty Acids on Hormonal Profile and Ovarian Stromal Blood Flow in Patients with Polycystic Ovary Syndrome,” Evidence Based Women’s Health Journal 9, no. 4 (November 1, 2019): 542–48, https://doi.org/10.21608/ebwhj.2019.64357.↩︎
Bogna Grygiel-Górniak, “Peroxisome Proliferator-Activated Receptors and Their Ligands: Nutritional and Clinical Implications – a Review,” Nutrition Journal 13, no. 1 (December 2014): 17, https://doi.org/10.1186/1475-2891-13-17.↩︎
Elahe Mohammadi and Maryam Rafraf, “Benefits of Omega-3 Fatty Acids Supplementation on Serum Paraoxonase 1 Activity and Lipids Ratios in Polycystic Ovary Syndrome,” text/html, Health Promotion Perspectives; ISSN: 2228-6497, 2012, https://doi.org/10.5681/HPP.2012.023; Kailin Yang et al., “Effectiveness of Omega-3 Fatty Acid for Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis,” Reproductive Biology and Endocrinology 16, no. 1 (December 2018): 27, https://doi.org/10.1186/s12958-018-0346-x.↩︎
Yang et al., “Effectiveness of Omega-3 Fatty Acid for Polycystic Ovary Syndrome,” December 2018.↩︎
Philip C. Calder, “Omega-3 Fatty Acids and Inflammatory Processes,” Nutrients 2, no. 3 (March 18, 2010): 355–74, https://doi.org/10.3390/nu2030355.↩︎
Philip C. Calder, “Omega‐3 Polyunsaturated Fatty Acids and Inflammatory Processes: Nutrition or Pharmacology?,” British Journal of Clinical Pharmacology 75, no. 3 (March 2013): 645–62, https://doi.org/10.1111/j.1365-2125.2012.04374.x.↩︎
Calder.↩︎
Inês Ferreira et al., “Resolvins, Protectins, and Maresins: DHA-Derived Specialized Pro-Resolving Mediators, Biosynthetic Pathways, Synthetic Approaches, and Their Role in Inflammation,” Molecules 27, no. 5 (March 3, 2022): 1677, https://doi.org/10.3390/molecules27051677.↩︎
Grygiel-Górniak, “Peroxisome Proliferator-Activated Receptors and Their Ligands.”↩︎
Avijit Kar et al., “Omega-3 Fatty Acids Mediated Cellular Signaling and Its Regulation in Human Health,” Clinical Nutrition Open Science 52 (December 2023): 72–86, https://doi.org/10.1016/j.nutos.2023.10.004.↩︎
Maarouf et al., “Effect of Omega-3 Fatty Acids on Hormonal Profile and Ovarian Stromal Blood Flow in Patients with Polycystic Ovary Syndrome.”↩︎
Fiza Komal et al., “Impact of Different Omega-3 Fatty Acid Sources on Lipid, Hormonal, Blood Glucose, Weight Gain and Histopathological Damages Profile in PCOS Rat Model,” Journal of Translational Medicine 18, no. 1 (December 2020): 349, https://doi.org/10.1186/s12967-020-02519-1.↩︎
Mohammadi and Rafraf, “Benefits of Omega-3 Fatty Acids Supplementation on Serum Paraoxonase 1 Activity and Lipids Ratios in Polycystic Ovary Syndrome.”↩︎
Yang et al., “Effectiveness of Omega-3 Fatty Acid for Polycystic Ovary Syndrome,” December 2018.↩︎
“Office of Dietary Supplements – Omega-3 Fatty Acids,” accessed November 25, 2024, https://ods.od.nih.gov/factsheets/Omega3FattyAcids-Consumer/.↩︎
Behnaz Khani, Farahnaz Mardanian, and SajadehJafari Fesharaki, “Omega-3 Supplementation Effects on Polycystic Ovary Syndrome Symptoms and Metabolic Syndrome,” Journal of Research in Medical Sciences 22, no. 1 (2017): 64, https://doi.org/10.4103/jrms.JRMS_644_16.↩︎
Kailin Yang et al., “Effectiveness of Omega-3 Fatty Acid for Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis,” Reproductive Biology and Endocrinology 16, no. 1 (December 2018): 27, https://doi.org/10.1186/s12958-018-0346-x.↩︎
“Office of Dietary Supplements – Omega-3 Fatty Acids.”↩︎
“Office of Dietary Supplements – Omega-3 Fatty Acids.”↩︎
“Office of Dietary Supplements – Omega-3 Fatty Acids.”↩︎
Kristina N. Krupa, Kristina Fritz, and Mayur Parmar, “Omega-3 Fatty Acids,” in StatPearls (Treasure Island (FL): StatPearls Publishing, 2025), http://www.ncbi.nlm.nih.gov/books/NBK564314/.↩︎
Mitchell S Buckley, Angela D Goff, and Walter E Knapp, “Fish Oil Interaction with Warfarin,” Annals of Pharmacotherapy 38, no. 1 (January 2004): 50–53, https://doi.org/10.1345/aph.1D007.↩︎
Datu Agasi Mohd Kamal et al., “Potential Health Benefits of Curcumin on Female Reproductive Disorders: A Review,” Nutrients 13, no. 9 (September 7, 2021): 3126, https://doi.org/10.3390/nu13093126.↩︎
Kamal et al.↩︎
Kamal et al.; Javad Heshmati et al., “Effects of Curcumin Supplementation on Blood Glucose, Insulin Resistance and Androgens in Patients with Polycystic Ovary Syndrome: A Randomized Double-Blind Placebo-Controlled Clinical Trial,” Phytomedicine 80 (January 2021): 153395, https://doi.org/10.1016/j.phymed.2020.153395.↩︎
Kamal et al., “Potential Health Benefits of Curcumin on Female Reproductive Disorders”; Heshmati et al., “Effects of Curcumin Supplementation on Blood Glucose, Insulin Resistance and Androgens in Patients with Polycystic Ovary Syndrome.”↩︎
Constanze Buhrmann et al., “Curcumin Modulates Nuclear Factor κB (NF-κB)-Mediated Inflammation in Human Tenocytes in Vitro,” Journal of Biological Chemistry 286, no. 32 (August 2011): 28556–66, https://doi.org/10.1074/jbc.M111.256180.↩︎
Karolina Jakubczyk et al., “Antioxidant Potential of Curcumin—A Meta-Analysis of Randomized Clinical Trials,” Antioxidants 9, no. 11 (November 6, 2020): 1092, https://doi.org/10.3390/antiox9111092.↩︎
Zeinab GHorbani, Azita Hekmatdoost, and Parvin Mirmiran, “Anti-Hyperglycemic and Insulin Sensitizer Effects of Turmeric and Its Principle Constituent Curcumin,” International Journal of Endocrinology and Metabolism 12, no. 4 (October 1, 2014), https://doi.org/10.5812/ijem.18081.↩︎
GHorbani, Hekmatdoost, and Mirmiran.↩︎
Wei Zhang et al., “The Therapeutic Effects of Curcumin on Polycystic Ovary Syndrome by Upregulating PPAR-γ Expression and Reducing Oxidative Stress in a Rat Model,” Frontiers in Endocrinology 15 (November 20, 2024): 1494852, https://doi.org/10.3389/fendo.2024.1494852.↩︎
Qin Yang, Qin Wan, and Zhen Wang, “Curcumin Mitigates Polycystic Ovary Syndrome in Mice by Suppressing TLR4/MyD88/NF-κB Signaling Pathway Activation and Reducing Intestinal Mucosal Permeability,” Scientific Reports 14, no. 1 (December 2, 2024): 29848, https://doi.org/10.1038/s41598-024-81034-5.↩︎
Sabiha Ataç Asan et al., “The Effects of Curcumin Supplementation Added to Diet on Anthropometric and Biochemical Status in Women with Polycystic Ovary Syndrome: A Randomized, Placebo-Controlled Trial,” Progress in Nutrition 22, no. 4 (January 11, 2021): e2020089, https://doi.org/10.23751/pn.v22i4.10460.↩︎
Yang, Wan, and Wang, “Curcumin Mitigates Polycystic Ovary Syndrome in Mice by Suppressing TLR4/MyD88/NF-κB Signaling Pathway Activation and Reducing Intestinal Mucosal Permeability.”↩︎
Yung-Jiun Chien et al., “Effects of Curcumin on Glycemic Control and Lipid Profile in Polycystic Ovary Syndrome: Systematic Review with Meta-Analysis and Trial Sequential Analysis,” Nutrients 13, no. 2 (February 21, 2021): 684, https://doi.org/10.3390/nu13020684.↩︎
Bharat B. Aggarwal and Kuzhuvelil B. Harikumar, “Potential Therapeutic Effects of Curcumin, the Anti-Inflammatory Agent, against Neurodegenerative, Cardiovascular, Pulmonary, Metabolic, Autoimmune and Neoplastic Diseases,” The International Journal of Biochemistry & Cell Biology 41, no. 1 (January 2009): 40–59, https://doi.org/10.1016/j.biocel.2008.06.010.↩︎
Ashika Ajitkumar Ashika Ajitkumar et al., “Drug-Induced Liver Injury Secondary to Turmeric Use,” European Journal of Case Reports in Internal Medicine, April 21, 2023, https://doi.org/10.12890/2023_003845.↩︎
Basel Abdelazeem et al., “The Effects of Curcumin as Dietary Supplement for Patients with Polycystic Ovary Syndrome: An Updated Systematic Review and Meta‐analysis of Randomized Clinical Trials,” Phytotherapy Research 36, no. 1 (January 2022): 22–32, https://doi.org/10.1002/ptr.7274.↩︎
Sahdeo Prasad, Amit K. Tyagi, and Bharat B. Aggarwal, “Recent Developments in Delivery, Bioavailability, Absorption and Metabolism of Curcumin: The Golden Pigment from Golden Spice,” Cancer Research and Treatment 46, no. 1 (January 15, 2014): 2–18, https://doi.org/10.4143/crt.2014.46.1.2.↩︎
Abdelazeem et al., “The Effects of Curcumin as Dietary Supplement for Patients with Polycystic Ovary Syndrome.”↩︎
El-Saadony et al., “Impacts of Turmeric and Its Principal Bioactive Curcumin on Human Health.”↩︎
Susan Hewlings and Douglas Kalman, “Curcumin: A Review of Its Effects on Human Health,” Foods 6, no. 10 (October 22, 2017): 92, https://doi.org/10.3390/foods6100092.↩︎
Mohamed T. El-Saadony et al., “Impacts of Turmeric and Its Principal Bioactive Curcumin on Human Health: Pharmaceutical, Medicinal, and Food Applications: A Comprehensive Review,” Frontiers in Nutrition 9 (January 10, 2023): 1040259, https://doi.org/10.3389/fnut.2022.1040259.↩︎
Tiziana Filardi et al., “Curcumin: Could This Compound Be Useful in Pregnancy and Pregnancy-Related Complications?,” Nutrients 12, no. 10 (October 17, 2020): 3179, https://doi.org/10.3390/nu12103179.↩︎
S.A. Lee and M.R. Bedford, “Inositol – An Effective Growth Promotor?,” World’s Poultry Science Journal 72, no. 4 (December 1, 2016): 743–60, https://doi.org/10.1017/S0043933916000660.↩︎
George M. Kapalka, “Anxiety Disorders,” in Nutritional and Herbal Therapies for Children and Adolescents (Elsevier, 2010), 219–58, https://doi.org/10.1016/B978-0-12-374927-7.00008-X; Lee and Bedford, “Inositol – An Effective Growth Promotor?”↩︎
Dhani Raj Chhetri, “Myo-Inositol and Its Derivatives: Their Emerging Role in the Treatment of Human Diseases,” Frontiers in Pharmacology 10 (October 11, 2019): 1172, https://doi.org/10.3389/fphar.2019.01172.↩︎
Becky Tu-Sekine and Sangwon F. Kim, “The Inositol Phosphate System—A Coordinator of Metabolic Adaptability,” International Journal of Molecular Sciences 23, no. 12 (June 16, 2022): 6747, https://doi.org/10.3390/ijms23126747.↩︎
Tu-Sekine and Kim; David O. Norris and James A. Carr, “Synthesis, Metabolism, and Actions of Bioregulators,” in Vertebrate Endocrinology (Elsevier, 2013), 41–91, https://doi.org/10.1016/B978-0-12-394815-1.00003-3.↩︎
Arturo Bevilacqua and Mariano Bizzarri, “Inositols in Insulin Signaling and Glucose Metabolism,” International Journal of Endocrinology 2018 (November 25, 2018): 1–8, https://doi.org/10.1155/2018/1968450.↩︎
James J DiNicolantonio and James H O’Keefe, “Myo-Inositol for Insulin Resistance, Metabolic Syndrome, Polycystic Ovary Syndrome and Gestational Diabetes,” Open Heart 9, no. 1 (March 2022): e001989, https://doi.org/10.1136/openhrt-2022-001989.↩︎
Carmen Concerto et al., “Neurobiology and Applications of Inositol in Psychiatry: A Narrative Review,” Current Issues in Molecular Biology 45, no. 2 (February 20, 2023): 1762–78, https://doi.org/10.3390/cimb45020113.↩︎
Joseph Levine, “Controlled Trials of Inositol in Psychiatry,” European Neuropsychopharmacology 7, no. 2 (May 1997): 147–55, https://doi.org/10.1016/S0924-977X(97)00409-4.↩︎
Tonino Cantelmi and Cherubino Di Lorenzo, “Myo-Inositol Could Restore Peripheral Inositol Depletion Induced by Treatments for Psychiatric and Neurological Conditions,” in A Clinical Guide to Inositols (Elsevier, 2023), 73–85, https://doi.org/10.1016/B978-0-323-91673-8.00003-0.↩︎
Vittorio Unfer et al., “Myo-Inositol Effects in Women with PCOS: A Meta-Analysis of Randomized Controlled Trials,” Endocrine Connections 6, no. 8 (November 2017): 647–58, https://doi.org/10.1530/EC-17-0243.↩︎
Philippe Merviel et al., “Impact of Myo-Inositol Treatment in Women with Polycystic Ovary Syndrome in Assisted Reproductive Technologies,” Reproductive Health 18, no. 1 (December 2021): 13, https://doi.org/10.1186/s12978-021-01073-3.↩︎
Victoria Fitz et al., “Inositol for Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis to Inform the 2023 Update of the International Evidence-Based PCOS Guidelines,” The Journal of Clinical Endocrinology & Metabolism 109, no. 6 (May 17, 2024): 1630–55, https://doi.org/10.1210/clinem/dgad762.↩︎
Dorina Greff et al., “Inositol Is an Effective and Safe Treatment in Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis of Randomized Controlled Trials,” Reproductive Biology and Endocrinology 21, no. 1 (January 26, 2023): 10, https://doi.org/10.1186/s12958-023-01055-z.↩︎
Marian G Showell et al., “Inositol for Subfertile Women with Polycystic Ovary Syndrome,” ed. Cochrane Gynaecology and Fertility Group, Cochrane Database of Systematic Reviews 2018, no. 12 (December 20, 2018), https://doi.org/10.1002/14651858.CD012378.pub2.↩︎
PubChem, “Berberine,” accessed March 3, 2025, https://pubchem.ncbi.nlm.nih.gov/compound/2353; Maria A. Neag et al., “Berberine: Botanical Occurrence, Traditional Uses, Extraction Methods, and Relevance in Cardiovascular, Metabolic, Hepatic, and Renal Disorders,” Frontiers in Pharmacology 9 (August 21, 2018): 557, https://doi.org/10.3389/fphar.2018.00557.↩︎
Weidong Li et al., “Berberine Regulates AMP‐activated Protein Kinase Signaling Pathways and Inhibits Colon Tumorigenesis in Mice,” Molecular Carcinogenesis 54, no. 10 (October 2015): 1096–1109, https://doi.org/10.1002/mc.22179.↩︎
Alexandra Cok et al., “Berberine Acutely Activates the Glucose Transport Activity of GLUT1,” Biochimie 93, no. 7 (July 2011): 1187–92, https://doi.org/10.1016/j.biochi.2011.04.013.↩︎
Li et al., “Berberine Regulates AMP‐activated Protein Kinase Signaling Pathways and Inhibits Colon Tumorigenesis in Mice”; Suyesh Shrivastava et al., “Addressing the Preventive and Therapeutic Perspective of Berberine against Diabetes,” Heliyon 9, no. 11 (November 2023): e21233, https://doi.org/10.1016/j.heliyon.2023.e21233.↩︎
Ayudiah Rizki Utami, Iman Permana Maksum, and Yusi Deawati, “Berberine and Its Study as an Antidiabetic Compound,” Biology 12, no. 7 (July 8, 2023): 973, https://doi.org/10.3390/biology12070973.↩︎
Utami, Maksum, and Deawati; Yongqing Yang et al., “Berberine Induces Lipolysis in Porcine Adipocytes by Activating the AMP‑activated Protein Kinase Pathway,” Molecular Medicine Reports, April 10, 2020, https://doi.org/10.3892/mmr.2020.11070.↩︎
Yang et al., “Berberine Induces Lipolysis in Porcine Adipocytes by Activating the AMP‑activated Protein Kinase Pathway.”↩︎
Jun Yin et al., “Berberine Improves Glucose Metabolism through Induction of Glycolysis,” American Journal of Physiology-Endocrinology and Metabolism 294, no. 1 (January 2008): E148–56, https://doi.org/10.1152/ajpendo.00211.2007.↩︎
Yin et al.; Ru Feng et al., “Transforming Berberine into Its Intestine-Absorbable Form by the Gut Microbiota,” Scientific Reports 5, no. 1 (July 15, 2015): 12155, https://doi.org/10.1038/srep12155.↩︎
Feng et al., “Transforming Berberine into Its Intestine-Absorbable Form by the Gut Microbiota.”↩︎
Neha Mishra, Ruchi Verma, and Payal Jadaun, “Study on the Effect of Berberine, Myoinositol, and Metformin in Women with Polycystic Ovary Syndrome: A Prospective Randomised Study,” Cureus, January 31, 2022, https://doi.org/10.7759/cureus.21781.↩︎
M. Rondanelli et al., “Polycystic Ovary Syndrome Management: A Review of the Possible Amazing Role of Berberine,” Archives of Gynecology and Obstetrics 301, no. 1 (January 2020): 53–60, https://doi.org/10.1007/s00404-020-05450-4; Mariangela Rondanelli et al., “Berberine Phospholipid Is an Effective Insulin Sensitizer and Improves Metabolic and Hormonal Disorders in Women with Polycystic Ovary Syndrome: A One-Group Pretest–Post-Test Explanatory Study,” Nutrients 13, no. 10 (October 19, 2021): 3665, https://doi.org/10.3390/nu13103665.↩︎
Xiao-Ke Wu et al., “Randomized Controlled Trial of Letrozole, Berberine, or a Combination for Infertility in the Polycystic Ovary Syndrome,” Fertility and Sterility 106, no. 3 (September 2016): 757-765.e1, https://doi.org/10.1016/j.fertnstert.2016.05.022.↩︎
Rondanelli et al., “Berberine Phospholipid Is an Effective Insulin Sensitizer and Improves Metabolic and Hormonal Disorders in Women with Polycystic Ovary Syndrome.”↩︎
Meng-Fei Li, Xiao-Meng Zhou, and Xue-Lian Li, “The Effect of Berberine on Polycystic Ovary Syndrome Patients with Insulin Resistance (PCOS‐IR): A Meta‐Analysis and Systematic Review,” ed. I-Min Liu, Evidence-Based Complementary and Alternative Medicine 2018, no. 1 (January 2018): 2532935, https://doi.org/10.1155/2018/2532935.↩︎
Mishra, Verma, and Jadaun, “Study on the Effect of Berberine, Myoinositol, and Metformin in Women with Polycystic Ovary Syndrome.”↩︎
Li, Zhou, and Li, “The Effect of Berberine on Polycystic Ovary Syndrome Patients with Insulin Resistance (PCOS‐IR).”↩︎
Rondanelli et al., “Polycystic Ovary Syndrome Management”; Li, Zhou, and Li, “The Effect of Berberine on Polycystic Ovary Syndrome Patients with Insulin Resistance (PCOS‐IR).”↩︎
Wenting Xie et al., “Glucose-Lowering Effect of Berberine on Type 2 Diabetes: A Systematic Review and Meta-Analysis,” Frontiers in Pharmacology 13 (November 16, 2022): 1015045, https://doi.org/10.3389/fphar.2022.1015045.↩︎
Lichao Zhang et al., “Effects of Berberine on the Gastrointestinal Microbiota,” Frontiers in Cellular and Infection Microbiology 10 (February 19, 2021): 588517, https://doi.org/10.3389/fcimb.2020.588517.↩︎
Ryan S. Funk et al., “Variability in Potency Among Commercial Preparations of Berberine,” Journal of Dietary Supplements 15, no. 3 (May 4, 2018): 343–51, https://doi.org/10.1080/19390211.2017.1347227.↩︎
Nestoras Mathioudakis, “A Berberine Derivative for Treatment of Type 2 Diabetes,” JAMA Network Open 8, no. 3 (March 3, 2025): e2462195, https://doi.org/10.1001/jamanetworkopen.2024.62195.↩︎
Mathioudakis; Ying Guo et al., “Repeated Administration of Berberine Inhibits Cytochromes P450 in Humans,” European Journal of Clinical Pharmacology 68, no. 2 (February 2012): 213–17, https://doi.org/10.1007/s00228-011-1108-2.↩︎
“Berberine,” in Mother To Baby | Fact Sheets (Brentwood (TN): Organization of Teratology Information Specialists (OTIS), 1994), http://www.ncbi.nlm.nih.gov/books/NBK600384/.↩︎
Peres et al., “The Health Effects of Low Glycemic Index and Low Glycemic Load Interventions on Prediabetes and Type 2 Diabetes Mellitus”; Rizkalla, Bellisle, and Slama, “Health Benefits of Low Glycaemic Index Foods, Such as Pulses, in Diabetic Patients and Healthy Individuals.”↩︎
Clar et al., “Low Glycaemic Index Diets for the Prevention of Cardiovascular Disease”; Chiu and Taylor, “Dietary Hyperglycemia, Glycemic Index and Metabolic Retinal Diseases.”↩︎
Peres et al., “The Health Effects of Low Glycemic Index and Low Glycemic Load Interventions on Prediabetes and Type 2 Diabetes Mellitus”; Rizkalla, Bellisle, and Slama, “Health Benefits of Low Glycaemic Index Foods, Such as Pulses, in Diabetic Patients and Healthy Individuals.”↩︎
Manta et al., “Glycemic Index and Glycemic Load Estimates in the Dietary Approach of Polycystic Ovary Syndrome”; Chang et al., “Low Glycemic Load Experimental Diet More Satiating Than High Glycemic Load Diet.”↩︎
Kaur et al., “The Impact of a Low Glycaemic Index (GI) Diet on Simultaneous Measurements of Blood Glucose and Fat Oxidation.”↩︎
Marsh et al., “Effect of a Low Glycemic Index Compared with a Conventional Healthy Diet on Polycystic Ovary Syndrome.”↩︎
Farnaz Shishehgar et al., “Does a Restricted Energy Low Glycemic Index Diet Have a Different Effect on Overweight Women with or without Polycystic Ovary Syndrome?,” BMC Endocrine Disorders 19, no. 1 (December 2019): 93, https://doi.org/10.1186/s12902-019-0420-1.↩︎
Wang et al., “Effects of High-Protein Diets on the Cardiometabolic Factors and Reproductive Hormones of Women with Polycystic Ovary Syndrome.”↩︎
Sørensen et al., “Effects of Increased Dietary Protein-to-Carbohydrate Ratios in Women with Polycystic Ovary Syndrome.”↩︎
Lisa J. Moran et al., “Dietary Composition in the Treatment of Polycystic Ovary Syndrome: A Systematic Review to Inform Evidence-Based Guidelines,” Journal of the Academy of Nutrition and Dietetics 113, no. 4 (April 2013): 520–45, https://doi.org/10.1016/j.jand.2012.11.018.↩︎
Luigi Barrea et al., “Adherence to the Mediterranean Diet, Dietary Patterns and Body Composition in Women with Polycystic Ovary Syndrome (PCOS),” Nutrients 11, no. 10 (September 23, 2019): 2278, https://doi.org/10.3390/nu11102278.↩︎
Wajeed Masood et al., “Ketogenic Diet,” in StatPearls (Treasure Island (FL): StatPearls Publishing, 2025), http://www.ncbi.nlm.nih.gov/books/NBK499830/.↩︎
John C Mavropoulos et al., “The Effects of a Low-Carbohydrate, Ketogenic Diet on the Polycystic Ovary Syndrome: A Pilot Study,” Nutrition & Metabolism 2, no. 1 (December 2005): 35, https://doi.org/10.1186/1743-7075-2-35; Antonio Paoli et al., “Effects of a Ketogenic Diet in Overweight Women with Polycystic Ovary Syndrome,” Journal of Translational Medicine 18, no. 1 (December 2020): 104, https://doi.org/10.1186/s12967-020-02277-0.↩︎
Mavropoulos et al., “The Effects of a Low-Carbohydrate, Ketogenic Diet on the Polycystic Ovary Syndrome.”↩︎
Sørensen et al., “Effects of Increased Dietary Protein-to-Carbohydrate Ratios in Women with Polycystic Ovary Syndrome.”↩︎
Wang et al., “Effects of High-Protein Diets on the Cardiometabolic Factors and Reproductive Hormones of Women with Polycystic Ovary Syndrome.”↩︎
Langyan et al., “Sustaining Protein Nutrition Through Plant-Based Foods.”↩︎
Joshua D. Brown et al., “Effects on Cardiovascular Risk Factors of Weight Losses Limited to 5–10 %,” Translational Behavioral Medicine 6, no. 3 (September 2016): 339–46, https://doi.org/10.1007/s13142-015-0353-9.↩︎
Simon Schenk et al., “Improved Insulin Sensitivity after Weight Loss and Exercise Training Is Mediated by a Reduction in Plasma Fatty Acid Mobilization, Not Enhanced Oxidative Capacity,” The Journal of Physiology 587, no. 20 (October 15, 2009): 4949–61, https://doi.org/10.1113/jphysiol.2009.175489; L D Clamp et al., “Enhanced Insulin Sensitivity in Successful, Long-Term Weight Loss Maintainers Compared with Matched Controls with No Weight Loss History,” Nutrition & Diabetes 7, no. 6 (June 19, 2017): e282–e282, https://doi.org/10.1038/nutd.2017.31.↩︎
Lei Zheng et al., “Obesity and Its Impact on Female Reproductive Health: Unraveling the Connections,” Frontiers in Endocrinology 14 (January 9, 2024): 1326546, https://doi.org/10.3389/fendo.2023.1326546; Grei Shele, Jessica Genkil, and Diana Speelman, “A Systematic Review of the Effects of Exercise on Hormones in Women with Polycystic Ovary Syndrome,” Journal of Functional Morphology and Kinesiology 5, no. 2 (May 31, 2020): 35, https://doi.org/10.3390/jfmk5020035; Luigi Barrea et al., “PCOS and Nutritional Approaches: Differences between Lean and Obese Phenotype,” Metabolism Open 12 (December 2021): 100123, https://doi.org/10.1016/j.metop.2021.100123.↩︎
Josefin Kataoka et al., “Effects of Weight Loss Intervention on Anxiety, Depression and Quality of Life in Women with Severe Obesity and Polycystic Ovary Syndrome,” Scientific Reports 14, no. 1 (June 12, 2024): 13495, https://doi.org/10.1038/s41598-024-63166-w; Carolyn Ee et al., “Providing Lifestyle Advice to Women with PCOS: An Overview of Practical Issues Affecting Success,” BMC Endocrine Disorders 21, no. 1 (November 23, 2021): 234, https://doi.org/10.1186/s12902-021-00890-8.↩︎
Schenk et al., “Improved Insulin Sensitivity after Weight Loss and Exercise Training Is Mediated by a Reduction in Plasma Fatty Acid Mobilization, Not Enhanced Oxidative Capacity.”↩︎
Shele, Genkil, and Speelman, “A Systematic Review of the Effects of Exercise on Hormones in Women with Polycystic Ovary Syndrome.”↩︎
Angelo Sabag et al., “Exercise in the Management of Polycystic Ovary Syndrome: A Position Statement from Exercise and Sports Science Australia,” Journal of Science and Medicine in Sport 27, no. 10 (October 2024): 668–77, https://doi.org/10.1016/j.jsams.2024.05.015.↩︎
Sabag et al.; Upasana Pandit et al., “The Effect of Exercise Training on Body Composition, Insulin Resistance and High Sensitivity C-Reactive Protein (Hs-CRP) in Women With Polycystic Ovary Syndrome: A Pilot Study From North India,” Cureus, April 9, 2022, https://doi.org/10.7759/cureus.23994; Leanne M. Redman, Karen Elkind-Hirsch, and Eric Ravussin, “Aerobic Exercise in Women with Polycystic Ovary Syndrome Improves Ovarian Morphology Independent of Changes in Body Composition,” Fertility and Sterility 95, no. 8 (June 2011): 2696–99, https://doi.org/10.1016/j.fertnstert.2011.01.137.↩︎
Somayeh Mohammadi, Amirabbas Monazzami, and Solmaz Alavimilani, “Effects of Eight-Week High-Intensity Interval Training on Some Metabolic, Hormonal and Cardiovascular Indices in Women with PCOS: A Randomized Controlled Trail,” BMC Sports Science, Medicine and Rehabilitation 15, no. 1 (March 29, 2023): 47, https://doi.org/10.1186/s13102-023-00653-z; Ling Jiang, Yaxin Chen, and Meiying Huang, “The Impact of High-Intensity Interval Training on Insulin Sensitivity and Quality of Life in Women with Overweight Polycystic Ovary Syndrome,” Technology and Health Care 33, no. 2 (March 2025): 1038–45, https://doi.org/10.1177/09287329241296228.↩︎
Paraskevi Pericleous and Savvas Stephanides, “Can Resistance Training Improve the Symptoms of Polycystic Ovary Syndrome?,” BMJ Open Sport & Exercise Medicine 4, no. 1 (August 2018): e000372, https://doi.org/10.1136/bmjsem-2018-000372.↩︎
Zahra Salajegheh et al., “Mindfulness-Based Stress Reduction (MBSR) Effects on the Worries of Women with Poly Cystic Ovary Syndrome (PCOS),” BMC Psychiatry 23, no. 1 (March 21, 2023): 185, https://doi.org/10.1186/s12888-023-04671-6.↩︎

